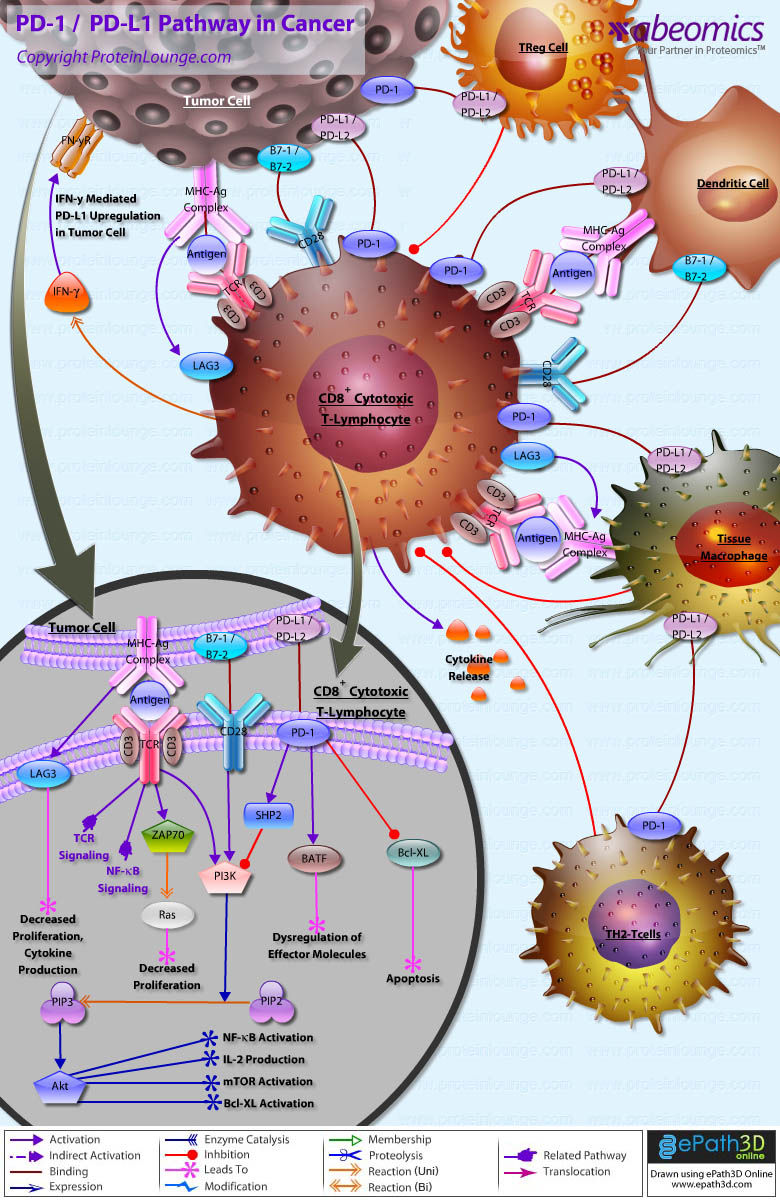
PD-1/PD-L1 Pathway in Cancer
Immune checkpoints refer to a plethora of inhibitory pathways hardwired into the immune system that are crucial for maintaining self-tolerance and limiting collateral tissue damage during anti-microbial immune responses. Checkpoint molecules include Cytotoxic T Lymphocyte Antigen-4 (CTLA-4), Programmed Death-1 (PD-1), Lymphocyte Activation Gene-3 (LAG-3), and T-cell immunoglobulin and Mucin protein-3 (TIM-3) as well as several others (Ref.1). PD-1, an immunoinhibitory receptor of the CD28 family, is a type 1 trans-membrane protein that encoded by the PDCD1 gene. The structure of the PD-1 includes an extracellular IgV domain, a hydrophobic trans-membrane region and an intracellular domain. PD-1 has two ligands: PD-L1 (also named B7-H1; CD274) and PD-L2 (B7-DC; CD273), that are both co-inhibitory (Ref.2 and 3). PD-1 is inducibly expressed by T cells and B cells after activation as well as natural killer T (NKT) cells, NK cells, activated monocytes and some subsets of dendritic cells (DCs). PD-1 is upregulated after TCR or BCR engagement on naïve lymphocytes and persistent antigen stimulation maintains high PD-1 expression. The common γ-chain cytokines (IL-2, IL-7, IL-15 and IL-21), TLRs and interferons also can potentiate PD-1 expression on T cells. NFATc1 together with AP-1 and NF-kB constitute the most critical transcription factors activated upon antigen recognition by T cells. PD-1 transduces an inhibitory signal when it is bound by its ligands in the presence of TCR or BCR activation. Phosphorylation of the ITSM motif of PD-1 leads to the recruitment of SH2-domain containing tyrosine phosphatase 2 (SHP-2), and possibly SHP-1, to the cytoplasmic domain of PD-1, which then down-regulates CD28-mediated PI3K activity and consequently, leads to less activation of Akt. PD-1 ligation also inhibits the phosphorylation of other signaling molecules including CD3, ZAP70 and PCK (Ref.4).
The PD-1/PD-L1 interaction inhibits T lymphocyte proliferation, survival and effector functions (cytotoxicity, cytokine release), induces apoptosis of tumor-specific T cells, promotes the differentiation of CD4+ T cells into Foxp3+ regulatory T cells, as well as the resistance of tumor cells to CTL attack. PD-L1 is expressed on resting T cells, B cells, dendritic cells, macrophage, vascular endothelial cells and pancreatic islet cells. PD-L2 expression is seen on macrophages and dendritic cells alone and is far less prevalent than PD-L1 across tumor types. PD-L1 is also expressed in various types of cancers, especially in NSCLC, melanoma, renal cell carcinoma, gastric cancer, hepatocellular as well as cutaneous and various leukemias, multiple myeloma and so on (Ref.2 and 3).
The present research of immune checkpoint molecules are mainly focus on cytotoxic T lymphocyte-associated antigen 4 (CLTA-4), Programmed death-1 (PD-1) and its ligands PD-L1 (B7H1) and PD-L2 (B7-DC). CTLA-4 regulates T cell activity in the early stage predominantly, and PD-1 mainly limits the activity of T-cell in the tumor microenvironment at later stage of tumor growth. Utilizing the immune checkpoint blockers to block the interactions between PD-1 and its ligands has shown benefits in clinical trials. PD-1 and its ligands have been rapidly established as the currently most important breakthrough targets in the development of effective immunotherapy (Ref.5).
References:
1. The blockade of immune checkpoints in cancer immunotherapy.
Pardoll DM.
Nat Rev Cancer. 2012 Mar 22;12(4):252-64. doi: 10.1038/nrc3239. Review.
2. Development of PD-1/PD-L1 Pathway in Tumor Immune Microenvironment and Treatment for Non-Small Cell Lung Cancer.
He J, Hu Y, Hu M, Li B.
Sci Rep. 2015 Aug 17;5:13110. doi: 10.1038/srep13110. Review.
3. Targeting PD-1/PD-L1 interactions for cancer immunotherapy.
Zitvogel L, Kroemer G.
Oncoimmunology. 2012 Nov 1;1(8):1223-1225.
4. Role of the PD-1 Pathway in the Immune Response
Leonardo V. Riella, Alison M. Paterson, Arlene H. Sharpe, Anil Chandraker
Am J Transplant. Author manuscript; available in PMC 2013 Sep 26.
Published in final edited form as: Am J Transplant. 2012 Oct; 12(10): 2575–2587. Published online 2012 Aug 17. doi: 10.1111/j.1600-6143.2012.04224.x
5. Molecular pathways: coexpression of immune checkpoint molecules: signaling pathways and implications for cancer immunotherapy.
Nirschl CJ, Drake CG.
Clin Cancer Res. 2013 Sep 15;19(18):4917-24. doi: 10.1158/1078-0432.CCR-12-1972. Review.
|