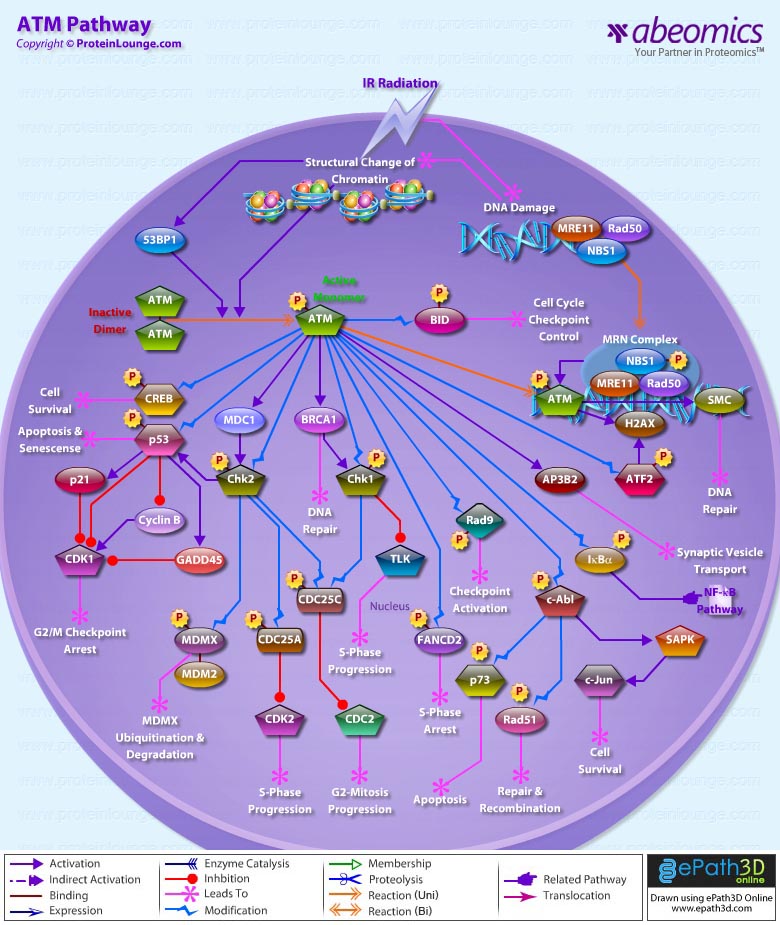
ATM (Ataxia Telangiectasia Mutated Protein) belongs to a family of Kinases that have sequence homology to PI3K (Phosphoinositide 3-Kinase). ATM is a key regulator of multiple signaling cascades which respond to DNA strand breaks induced by damaging agents IR (Ionizing Radiation), radiometric agents or by normal processes. These responses involve the activation of cell cycle Chk factors (Checkpoints factors), DNA repair and Apoptosis. In addition, ATM appears to function as a 'caretaker', suppressing tumorigenesis in specific T cell lineages. Its downstream targets include Chk1 (Cell Cycle Checkpoint Kinase-1), Chk2 (Cell Cycle Checkpoint Kinase-2), tumor suppressors like p53 and BRCA (Breast Cancer), DNA repair factors like Rad50, Rad51, GADD45 (Growth Arrest and DNA-Damage-inducible), and other signaling molecules like c-Abl and NF-KappaB (Nuclear Factor-Kappa B) (Ref.1). In non-irradiated cells ATM exists as a dimer, is not phosphorylated and is present throughout the nucleus. After irradiation, which leads to formation of DNA DSBs (DNA Double Strand Breaks), ATM becomes a monomer, it is phosphorylated on Ser1981 and some pool of it is present at sites of DNA DSBs. ATM monitors the presence of DNA DSBs indirectly, through DNA DSB-induced changes in chromatin structure. ATM is activated by the MRE11 (Meiotic Recombination-11)–Rad50–NBS1 (Nijmegen Breakage Syndrome-1) complex or 53BP1. The former is thought to be recruited at the DSB, whereas the latter is recruited at chromatin regions flanking the DNA DSB and extending up to a few megabases from the DSB. ATM phosphorylates NBS1 on several residues in response to DNA damage and a functional MRE11/Rad50/NBS1 complex is required for Chk2 activation. This links the MRE11/Rad50/NBS1 complex to DNA damage recognition. ATM activated at sites of DNA DSBs may also phosphorylate and activate ATM in the nucleoplasm (Ref.1 & 2).
Once ATM is activated it phosphorylates multiple substrates. Two of these, Chk2 and p53, mediate many of the cell cycle effects of ATM, while two others, SMC1 (Structural Maintainance of Chromosomes-1) and histone H2AX (H2A Histone family, member X), are important for cell survival after irradiation. Chk2 is a protein Kinase, that once activated amplifies the DNA damage signal of ATM. Two of the key substrates of Chk2 are CDC25A (Cell Division Cycle-25A) and CDC25C (Cell Division Cycle-25C). CDC25A and CDC25C are protein phosphatases; CDC25A activates CDK2 and promotes progression through S phase, while CDC25C activates CDC2 and promotes progression from G2 into mitosis. When CDC25A and CDC25C become phosphorylated by Chk2, their function is inhibited and cells delay progression through S phase or arrest in G2. Chk2 contributes to p53 regulation in part through phosphorylation of MDMX. The mechanism by which phosphorylation enhances MDMX–MDM2 (Mouse Double Minute-2) binding and MDMX ubiquitination by MDM2 remains to be further investigated. MDC1 (Mediator of DNA damage Checkpoint-1) localizes to sites of DNA breaks and associates with Chk2 after DNA damage. This association is mediated by the MDC1 FHA domain and the phosphorylated Thr 68 of Chk2. Cell cycle arrest in G1 is mediated by p53, which is a substrate of both Chk2 and ATM. p53 is a transcription factor that induces expression of p21 (Cyclin Dependent Kinase Inhibitor-p21)/Waf1, GADD45, and MDM2. p21 is a CDK(Cyclin-Dependent Kinase) inhibitor. p53 also inhibits G2-M transition by repressing the transcription of CDC2 (Cell Division Cycle-2, G1 to S and G2 to M) and Cyclin-B. p53 can also induce expression of genes that induce apoptosis; and in certain tissues, induction of p53 leads to apoptosis, rather than cell cycle arrest. Unlike, Chk2 and p53, Phosphorylated SMC1 and Histone H2AX are found exclusively at sites of DNA DSBs. SMC1 phosphorylation is critical for cells to survive after irradiation. Similar to SMC1 phosphorylation, histone H2AX phosphorylation also appears to be important for DNA repair. The mechanisms by which SMC1 and histone H2AX phosphorylation facilitate DNA repair are not known, but recent evidence suggests that phosphorylated histone H2AX recruits chromatin remodeling complexes to sites of DNA DSBs (Ref.3, 4 & 5).
ATM also interacts with the protein product of the c-Abl proto-oncogene. This non-receptor tyrosine Kinase is involved in several stress responses including activation of the SAPK (Stress-Activated Protein Kinase), Rad51 and p73. SAPK activation by c-Abl leads to activation of c-Jun and thus plays an important role in cell survival. c-Abl also phosphorylates Rad51, which is further involved in DNA repair and recombination processes. Activated c-Abl may also promote apoptosis via up-regulation of p73, a proapoptotic protein and a p53 homolog. ATM also interacts with AP3B2 (Adaptor-related Protein complex-3, Beta-2 subunit), a neuronal homolog of Beta-Adaptin thought to be involved in synaptic vesicle transport in neuronal cells. ATM phosphorylates ATF2 (Activating Transcription Factor-2) on Serine 490 and 498 following IR (Ionizing Radiation). Phosphorylation of ATF2 by ATM results in its rapid colocalization with Gamma-H2AX and MRN components into IRIF (IR-Induced Foci). ATM also phosphorylates IKappaB-Alpha and thus plays an important role in NF-KappaB activation. ATM also phosphorylates and activates Chk1, which phosphorylates TLK1 (Tousled Like Kinase-1). TLK activity is rapidly suppressed by DNA damage and by inhibitors of replication. Chk1 also phosphorylate a conserved site (Ser-216) on protein phosphatase CDC25C, which results in it being inactivated and bound by the 14-3-3 protein. The inactive CDC25C is then incapable of removing an inhibitory phosphate group on Tyr-15 of CDC2, preventing entry into mitosis. ATM also phosphorylates FANCD2 (Fanconi Anemia Complementation Group-D2) protein, which then leads to S phase arrest (Ref.5, 6 & 7).
BRCA1 is also activated by the protein kinase ATM that initiates cell cycle changes after DNA damage. BRCA1 physically interacts with Chk1 and stimulates its activity after DNA damage. Hyperphosphorylation of Rad9 induced by IR is also dependent on ATM. Ser(272) of Rad9 is phosphorylated directly by ATM. ATM also phosphorylates CREB (cAMP Response Element-Binding Protein-1) in vitro and in vivo in response to IR and H2O2 on a stress-inducible domain. ATM-mediated phosphorylation of CREB in response to DNA damage modulates CREB-dependent gene expression and dysregulation of the ATM-CREB pathway may contribute to neurodegeneration. ATM also phosphorylates BID (BH3 Interacting Domain Death Agonist). This phosphorylation is required for a downstream function in cell cycle checkpoint control. ATM also seems to have a role in telomere maintenance and replication. The disease AT (Ataxia-Telangiectasia), caused by mutations in the ATM gene, is an autosomal recessive disorder characterized by Progressive Cerebellar Ataxia, Cancer Predisposition, Oculocutaneous Telangiectasia and variable immune deficiencies. Inhibiting ATM might sensitize repair proficient cancer cells to radiation and chemotherapy. It is possible that targeting the ATM pathway might even introduce selectivity to current cancer therapy. However, the real test of this hypothesis will have to await the development of more potent and specific inhibitors for ATM (Ref.8, 9 & 10).
References
1.Exclusion/confirmation of Ataxia-telangiectasia via cell-cycle testing.
Heinrich T, Prowald C, Friedl R, Gottwald B, Kalb R, Neveling K, Herterich S, Hoehn H, Schindler D.
Eur J Pediatr. 2006 Jan 13;:1-8 [Epub ahead of print]
2.ATM signaling and 53BP1.
Zgheib O, Huyen Y, DiTullio RA Jr, Snyder A, Venere M, Stavridi ES, Halazonetis TD.
Radiother Oncol. 2005 Aug;76(2):119-22.
3.DNA damage regulates CHK2 association with chromatin.
Li J, Stern DF.
J Biol Chem. 2005 Sep 8; [Epub ahead of print]
4.ATM-mediated phosphorylations inhibit Mdmx/Mdm2 stabilization by HAUSP in favor of p53 activation.
Meulmeester E, Pereg Y, Shiloh Y, Jochemsen AG.
Cell Cycle. 2005 Sep;4(9):1166-70. Epub 2005 Sep 29.
5.Defective ATM-p53-mediated apoptotic pathway in multiple sclerosis.
Deng X, Ljunggren-Rose A, Maas K, Sriram S.
Ann Neurol. 2005 Oct;58(4):577-84.
6.ATM-dependent phosphorylation of ATF2 is required for the DNA damage response.
Bhoumik A, Takahashi S, Breitweiser W, Shiloh Y, Jones N, Ronai Z.
Mol Cell. 2005 May 27;18(5):577-87.
7.Suppression of Tousled-like kinase activity after DNA damage or replication block requires ATM, NBS1 and Chk1.
Krause DR, Jonnalagadda JC, Gatei MH, Sillje HH, Zhou BB, Nigg EA, Khanna K.
Oncogene. 2003 Sep 4;22(38):5927-37.
8.Direct regulation of CREB transcriptional activity by ATM in response to genotoxic stress.
Shi Y, Venkataraman SL, Dodson GE, Mabb AM, LeBlanc S, Tibbetts RS.
Proc Natl Acad Sci U S A. 2004 Apr 20;101(16):5898-903. Epub 2004 Apr 8.
9.Proapoptotic BID is an ATM effector in the DNA-damage response.
Kamer I, Sarig R, Zaltsman Y, Niv H, Oberkovitz G, Regev L, Haimovich G, Lerenthal Y, Marcellus RC, Gross A.
Cell. 2005 Aug 26;122(4):593-603.
10.ATM and ataxia telangiectasia.
McKinnon PJ.
EMBO Rep. 2004 Aug;5(8):772-6. Review.







