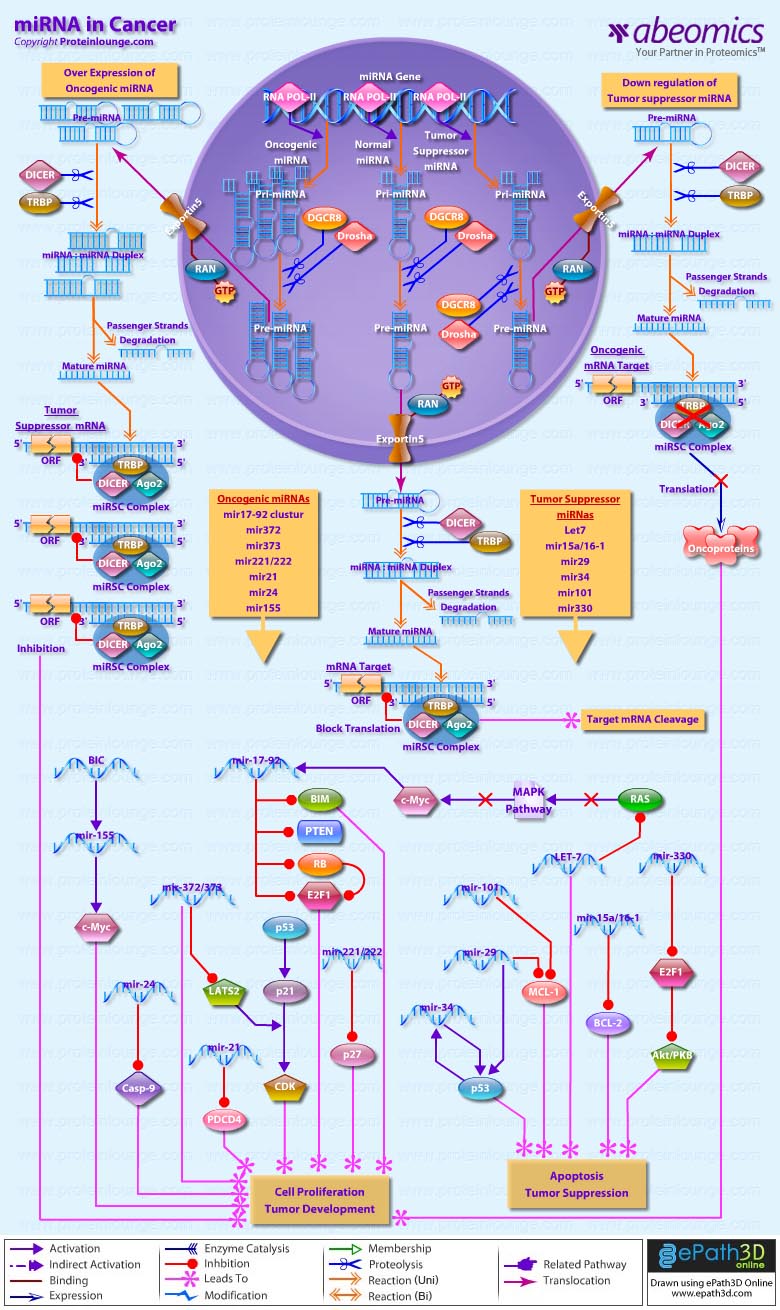
Over the last decade, a growing number of non-coding transcripts have been found to have roles in gene regulation and RNA processing. The most well known small non-coding RNAs are the miRNAs/miRs (microRNAs), which have been found to be involved in human tumorigenesis, revealing a new layer in the molecular architecture of cancer. Gene expression studies have shown that hundreds of miRNAs are deregulated in cancer cells, and functional studies have clarified that miRNAs are involved in all the molecular and biologic processes that drive tumorigenesis. miRNAs constitute a large class of phylogenetically conserved single-stranded RNA molecules of 19 to 25 nucleotides playing major role in post-transcriptional gene silencing by translational repression or mRNA cleavage (Ref. 1). Till date more than 500 miRNAs have been documented and the number is still growing. About 3% of human genes encode for miRNAs, and up to 30% of human protein coding genes are regulated by miRNAs (Ref. 2). Most miRNAs are transcribed by RNA Polymerase II as long pri-miRNAs (primary RNA) that contain a 5' CAP structure and a 3'polyadenylated tail. The pri-miRNAs are processed in the nucleus by the RNase III enzyme, Drosha, and the double-stranded- RNA-binding protein, Pasha (also known as DGCR8), into ~70-nucleotide pre-miRNAs, which fold into imperfect stem-loop structures. The pre-miRNAs are then exported into the cytoplasm by the RAN-GTP dependent transporter exportin 5 and undergo an additional processing step in which a double- stranded RNA of ~22 nucleotides in length, referred to as the miRNA:miRNA* duplex, is excised from the pre-miRNA hairpin by another RNAse III enzyme, Dicer, in association with its RNA binding partner TRBP. Subsequently, the miRNA:miRNA* duplex is incorporated into the miRISC (multiprotein RNA-induced silencing) complex, which includes the Argonaute proteins (Argonate2), where the passenger strand is removed and degraded and subsequently the mature miRNA guides the RISC to target mRNAs whose expression can be inhibited by translational repression through perfect or nearly perfect complementarity to 3' UTRs (Un-translated regions) or direct degradation of the target mRNA. It has been estimated that a single miRNA family can regulate as many as 200 different genes. Thus the effects of the miRNAs are pleotropic, and their aberrant expressions conceivably unbalance the cell¡¯s homeostasis, contributing to diseases, including cancer (Ref. 3 & 4).
miRNA expression correlates with various cancers, and these genes are thought to function as both tumor suppressors and oncogenes . About 50% of annotated human miRNAs are located in areas of the genome, known as fragile sites that are associated with cancer (Ref. 1). Expression of miRs and the role of miRs in cancer are tissue and tumor specific. Abnormal miR levels in tumors have important pathogenetic consequences: miRs may act as oncogenes or suppressor genes. miRs over expressed in tumors downregulate tumor suppressor genes, whereas miRs lost by tumors participate in oncogene overexpression (Ref. 5). In normal tissues, proper miRNA transcription, processing and binding to complementary sequences on the target mRNA results in the repression of target-gene expression through a block in protein translation or altered mRNA stability. The overall result is normal rates of cellular growth, proliferation, differentiation and cell death. The reduction or deletion of a miRNA that functions as a tumor suppressor leads to tumor formation. A reduction in or down regulation of mature miRNA levels can occur because of defects at any stage of miRNA biogenesis and ultimately leads to the inappropriate expression of the miRNA-target oncoprotein. The overall outcome might involve increased proliferation, invasiveness or angiogenesis, decreased levels of apoptosis, or undifferentiated or de-differentiated tissue, ultimately leading to tumor formation. The amplification or over expression of a miRNA that has an oncogenic role would also result in tumor formation. In this situation, increased amounts of a miRNA, which might be produced at inappropriate times or in the wrong tissues, would eliminate the expression of a miRNA-target tumor-suppressor gene and lead to cancer progression. Increased levels of mature miRNA might occur because of amplification of the miRNA gene, a constitutively active promoter, increased efficiency in miRNA processing or increased stability of the miRNA (Ref. 6).
Those miRNAs whose expression is increased in tumors are considered as oncogenes. These oncogene miRNAs, called ¡°oncomirs¡±, usually promote tumor development by negatively inhibiting tumor suppressor genes and/or genes that control cell differentiation or apoptosis (Ref. 7 & 6). mir-17¨C92 cluster is a classic example of an oncomir. This is a miRNA polycistron located at chromosome 13q31, a genomic locus that is amplified in lung cancer and several kinds of lymphoma, including diffuse large B-cell lymphoma. mir-17¨C92 cluster includes seven miRNAs: miR-17-5p, miR-17-3p, miR-18a, miR-19a, miR-20a, miR-19b-1 miR-92-1. Two tumor suppressor genes PTEN (phosphatase and tensin homolog deleted on chromosome ten) and RB2 are targeted by miR-17¨C92 cluster. PTEN promotes apoptosis through the P13K-Akt-PKB pathway. Over expression of mir-19 down regulates PTEN and leads to tumor development (Ref. 8, 9, 10 & 11). Unlike miR-19/PTEN axis toward apoptosis suppression, miR-17-92 increases proliferative capacity of Rb-deficient retinal cells. Deletion of Rb leads to compensatory up-regulation of the cyclin-dependent kinase inhibitor p21Cip1 and over expression of mir17-92 counteracts p21Cip1 up-regulation, promotes proliferation, and drive retinoblastoma formation. Over expression of mir-17¨C92 cluster also co-operates with c-Myc to accelerate lymphoma. Infect, miR-17-92 cluster itself is a transcriptional target of c-Myc. miR-17/20a targets E2F1, a regulator of the cell cycle and apoptosis. miR-92 targets BIM, a pro-apoptotic gene that counteracts the anti-apoptotic activity of genes such as Bcl2 (Ref. 9, 10 & 11). Another miRNA, miR-155, is linked with various lymphomas. This miRNA is encoded by a conserved region of the non-coding gene B- ell integration cluster, BIC, a common integration site for the avian leucosis virus, inducing B-cell lymphomas in collaboration with MYC (Ref. 3, 6 & 8). miR-372 and miR-373 are two additional examples of oncogenic miRNAs. These two miRNAs promote cell proliferation and tumor development by neutralizing p53-mediated CDK inhibition, possibly through direct inhibition of expression of the tumor suppressor gene LATS2 (Ref. 8 & 12). Another oncomir, mir-24, whose overexpression negatively regulates the proapoptotic factors caspase9 and APAF1, leading to cell proliferation and tumor development (Ref. 13 & 14). MiR-221 and miR-222 are two highly homologous microRNAs. These are positive regulators of prostate carcinoma growth through the repression of their target p27 which is a negative regulator of cell cycle progression (Ref. 15). In case of glioblastoma, over expression of oncogenic mir-21 inhibits its target programmed cell death 4 (Pdcd4), a known tumor-suppressor gene, and leads to increased proliferation of primary brain tumor cells (Ref. 16).
In oncogenesis, some miRNAs expression is decreased in cancerous cells. These types of miRNAs are considered tumor suppressor genes. Tumor suppressor miRNAs usually prevent tumor development by negatively inhibiting oncogenes and/or genes that control cell differentiation or apoptosis. Currently, several miRNAs are considered as tumor suppressor genes, for example, mir let-7, mir-15/16-1, mir-29, mir-34,mir-101& mir-330 (Ref. 8). let-7 is one of the founding members of the miRNA family. It is located at a chromosome region that is usually deleted in human cancers. let-7 is poorly expressed in lung cancers; reduced let-7 expression is significantly associated with shortened postoperative survival independent of disease stage. This confirms the tumor suppressor role of let-7. A recent study indicates that RAS oncogene is a direct target of let-7. let-7 negatively regulates RAS expression by pairing at the 3¡ä UTR of RAS mRNA for translational repression. Moreover let-7 negatively regulates the expression of RAS and MYC by targeting their mRNAs for translation repression (Ref. 8, 2 & 3). miRNAs encoded by the miR-15/16 cluster are also act as tumor suppressors. Expression of these miRNAs inhibits cell proliferation, promotes apoptosis of cancer cells, and suppresses tumorigenicity. Anti-apoptotic oncogene BCL2 is one of targets regulated by miR-15a and miR-16-1. Both these miRNAs negatively regulate BCL2 at a posttranscriptional level. Moreover, the Bcl2 repression by these miRNAs induces apoptosis in a chronic lymphocytic leukemia (Ref. 17 & 3). miR-101 and miR-29b exert their proapoptotic function via targeting Mcl-1,which is a member of Bcl-2 family and can inhibit apoptosis (Ref. 18 & 13).miR-29 also down-regulates p85a, a regulatory subunit of PI3K, and thereby enhances p53 activity through the negative loop between PI3K-AKT-MDM2 and p53 (Ref. 19). Another tumor suppressor miRNA, mir-34 promotes p53-mediated apoptosis, cell cycle arrest and senescence. Expression of miR-34 induces cell cycle arrest and thereby acts together with other effectors of the p53 tumor suppressor network to inhibit inappropriate cell proliferation. The expression of the mir-34 family members is regulated directly by p53, and consequently the expression of the mir-34 family reflects p53 activity (Ref. 2, 3, 19 & 13). microRNA mir-330 behaves like a potential tumor suppressor in many cancer types. It induces apoptosis of prostate cancer cells through E2F1- mediated suppression of Akt-phosphorylation (Ref. 18 & 20). Micro RNAs are causing tremendous excitement in cancer research. While the field of miRNAs and particularly the study of the roles of miRNAs in cancer is at an early stage, their potential as targeted therapeutic tools has not gone unnoticed. miRNA-based cancer therapies may be on the horizon in recent future.
References:
1. Non-coding RNAs and cancer: new paradigms in oncology.
Lee SK, Calin GA.
Discov Med. 2011 Mar;11(58):245-54.
2. MicroRNA: implications for cancer.
Sassen S, Miska EA, Caldas C.
Virchows Arch. 2008 Jan;452(1):1-10.
3. microRNAs and cancer: an overview.
Medina PP, Slack FJ.
Cell Cycle. 2008 Aug 15;7(16):2485-92.
4. The emerging roles of microRNAs in the molecular responses of metabolic rate depression.
Biggar KK, Storey KB.
J Mol Cell Biol. 2011 Jun;3(3):167-75.
5. MicroRNAs: a complex regulatory network drives the acquisition of malignant cell phenotype.
Santarpia L, Nicoloso M, Calin GA.
Endocr Relat Cancer. 2010 Jan 29;17(1):F51-75.
6. Oncomirs - microRNAs with a role in cancer.
Esquela-Kerscher A, Slack FJ.
Nat Rev Cancer. 2006 Apr;6(4):259-69.
7. Mechanisms and role of microRNA deregulation in cancer onset and progression.
Palmero EI, de Campos SG, Campos M, de Souza NC, Guerreiro ID, Carvalho AL, Marques MM.
Genet Mol Biol. 2011 Jul;34(3):363-70.
8. microRNAs as oncogenes and tumor suppressors.
Zhang B, Pan X, Cobb GP, Anderson TA.
Dev Biol. 2007 Feb 1;302(1):1-12.
9. Tumorigenicity of the miR-17-92 cluster distilled.
van Haaften G, Agami R.
Genes Dev. 2010 Jan 1;24(1):1-4.
10. miR-17~92 cooperates with RB pathway mutations to promote retinoblastoma.
Conkrite K, Sundby M, Mukai S, Thomson JM, Mu D, Hammond SM, MacPherson D.
Genes Dev. 2011 Aug 15;25(16):1734-45.
11. mir-17-92, a cluster of miRNAs in the midst of the cancer network.
Olive V, Jiang I, He L.
Int J Biochem Cell Biol. 2010 Aug;42(8):1348-54.
12. MicroRNA-372 is down-regulated and targets cyclin-dependent kinase 2 (CDK2) and cyclin A1 in human cervical cancer, which may contribute to tumorigenesis.
Tian RQ, Wang XH, Hou LJ, Jia WH, Yang Q, Li YX, Liu M, Li X, Tang H.
J Biol Chem. 2011 Jul 22;286(29):25556-63.
13. MicroRNA: A matter of life or death.
Wang Z.
World J Biol Chem. 2010 Apr 26;1(4):41-54.
14. microRNA-24a is required to repress apoptosis in the developing neural retina.
Walker JC, Harland RM.
Genes Dev. 2009 May 1;23(9):1046-51.
15. The inhibition of the highly expressed miR-221 and miR-222 impairs the growth of prostate carcinoma xenografts in mice.
Mercatelli N, Coppola V, Bonci D, Miele F, Costantini A, Guadagnoli M, Bonanno E, Muto G, Frajese GV, De Maria R, Spagnoli LG, Farace MG, Ciafrè SA.
PLoS One. 2008;3(12):e4029.
16. Downregulation of Pdcd4 by mir-21 facilitates glioblastoma proliferation in vivo.
Gaur AB, Holbeck SL, Colburn NH, Israel MA.
Neuro Oncol. 2011 Jun;13(6):580-90.
17. miR-15a and miR-16-1 in cancer: discovery, function and future perspectives.
Aqeilan RI, Calin GA, Croce CM.
Cell Death Differ. 2010 Feb;17(2):215-20.
18. MicroRNAs in cardiac apoptosis.
Li P.
J Cardiovasc Transl Res. 2010 Jun;3(3):219-24.
19. Tumor suppressor p53 meets microRNAs.
Feng Z, Zhang C, Wu R, Hu W.
J Mol Cell Biol. 2011 Feb;3(1):44-50.
20. MicroRNA-330 acts as tumor suppressor and induces apoptosis of prostate cancer cells through E2F1-mediated suppression of Akt phosphorylation.
Lee KH, Chen YL, Yeh SD, Hsiao M, Lin JT, Goan YG, Lu PJ.
Oncogene. 2009 Sep 24;28(38):3360-70.
|







