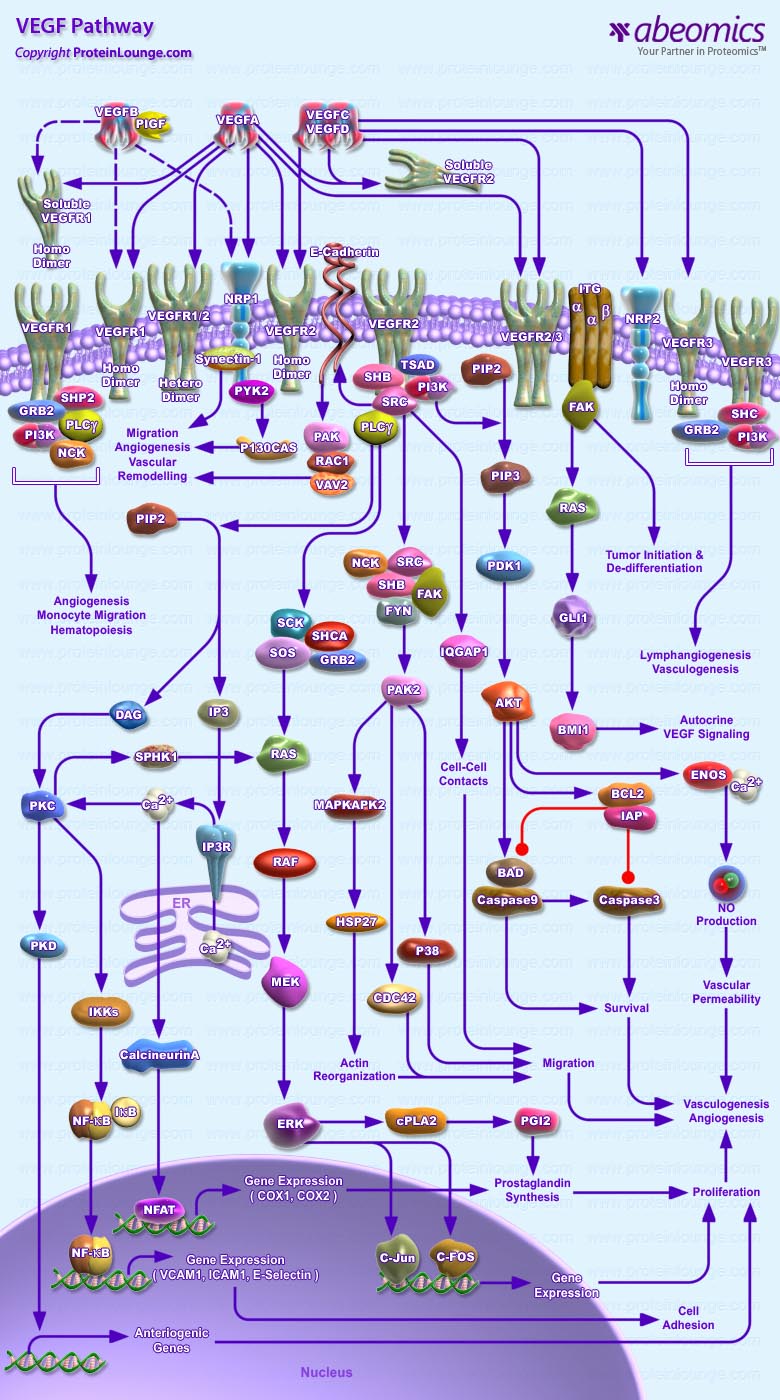
The vascular endothelial growth factor (VEGF) family of soluble protein growth factors is master regulators of in vasculogenesis and angiogenesis during blood vessel development. In mammals, the VEGF family consists of 5 members, VEGFA, B, C, and D and placenta growth factor (PIGF). VEGFs act through three structurally related VEGF receptor tyrosine kinases, denoted VEGFR1 (FLT1), VEGFR2 (FLK1), and VEGFR3 (FLT4). Binding of VEGF to its cognate VEGF receptor in cis or trans (e.g., by binding HS proteoglycans [HSPGs] on adjacent cells induces receptor homo- or heterodimerization (Ref.1 and 2). VEGFA binds VEGFR1 and VEGFR2. VEGFR2 is expressed mainly in endothelial cells, whereas VEGFR1 is expressed in endothelial cells as well as hematopoietic stem cells and inflammatory cells, such as monocytes and macrophages, in which it regulates chemotaxis. The VEGFR1 tyrosine kinase receptor (TKR) mediates various biologic effects of VEGFA, B, and PlGF. Potential interacting partners of VEGFR1 include Phospholipase C-gamma, PI3K, SHP2, growth factor receptor-bound protein 2 (GRB2), and NCK. VEGF or PlGF binding to VEGFR1 induces phosphorylation and activation of extracellular signal-regulated kinases 1 and 2 (ERK1/2) and p38 mitogen-activated protein kinase (MAPK) (Ref.3 and 4).
VEGFR2 (KDR, FLK1) is considered the major mediator of VEGFA, C, and D bioactivity in both physiologic and pathologic angiogenesis. In addition, VEGFR heterodimerization and interactions of VEGFR with co receptors such as neuropilins (NRP), heparan sulfate proteoglycans or ávâ3 integrin further expand the complexity of signaling pathways activated by VEGF and PlGF homo- or heterodimers. Phosphorylated VEGFR2 initiates downstream signaling pathways relevant to angiogenesis. VEGFA binding leads to the recruitment of molecules such as SH2 domain containing adaptor protein B (SHB), SRC proto-oncogene (SRC), SH2 domain containing 2 A (TSAD), fibroblast growth factor receptor substrate 2 (FRS2). An important pathway of VEGF regulated proliferation include PLC-gamma mediated activation of PKC and downstream induction of the ERK and other PKC-dependent pathways.VEGFR2 dependent activation of PI3K-AKT signaling regulates cell survival (Ref.5 and 6).
VEGFR3 is an orphan receptor for the VEGF receptors, VEGFR1 and VEGFR2. VEGFC and VEGFD have been identified as the ligands for VEGFR3. In addition to forming homodimers, VEGFR3 can also form complexes with VEGFR2. It plays important roles both in lymphangiogenesis and angiogenesis. Several downstream signal pathways including ERK1/2 and AKT are activated by VEGFR3. The AKT pathway was also shown to be important for survival, growth and migration of human microvascular endothelial cells. P70S6K and eNOS serve as downstream targets of the PI3K/AKT pathway in response to VEGFC (Ref.7 and 8). VEGFR signaling also involves coreceptor proteins neuropilin 1 and neuropilin 2.These transmembrane proteins are able to bind both VEGFs and semaphorins and regulate vascular and neural development (Ref. 7). VEGF does indeed serve multiple functions, including normal maintenance of endothelial and neural cell compartments. The VEGF-VEGFR system is crucial for tumour angiogenesis, and anti-VEGF-VEGFR molecules are now widely used in the clinical field to treat cancer patients (Ref.9).
References:
-
Stacker SA, Achen MG.
Chin J Cancer. 2013 Jun;32(6):297-302. doi: 10.5732/cjc.012.10319. Epub 2013 Feb 19.
-
Koch S, Claesson-Welsh L.
Cold Spring Harb Perspect Med. 2012 Jul;2(7):a006502. doi: 10.1101/cshperspect.a006502. Review.
-
Amano H, Kato S, Ito Y, Eshima K, Ogawa F, Takahashi R, Sekiguchi K, Tamaki H, Sakagami H, Shibuya M, Majima M.
PLoS One. 2015 Jul 2;10(7):e0131445. doi: 10.1371/journal.pone.0131445. eCollection 2015.
-
Kowanetz M, Ferrara N.
Clin Cancer Res. 2006 Sep 1;12(17):5018-22. Review.
-
Ohba T, Cates JM, Cole HA, Slosky DA, Haro H, Ando T, Schwartz HS, Schoenecker JG.
Mol Cancer Res. 2014 Aug;12(8):1100-11. doi: 10.1158/1541-7786.MCR-14-0037. Epub 2014 Apr 23.
-
Abhinand CS, Raju R, Soumya SJ, Arya PS, Sudhakaran PR.
J Cell Commun Signal. 2016 Dec;10(4):347-354. Epub 2016 Sep 12.
-
Deng Y, Zhang X, Simons M.
Arterioscler Thromb Vasc Biol. 2015 Feb;35(2):421-9. doi: 10.1161/ATVBAHA.114.304881. Epub 2014 Dec 18.
-
Coso S, Zeng Y, Opeskin K, Williams ED.
PLoS One. 2012;7(6):e39558. doi: 10.1371/journal.pone.0039558. Epub 2012 Jun 22.
-
Shibuya M.
J Biochem. 2013 Jan;153(1):13-9. doi: 10.1093/jb/mvs136. Epub 2012 Nov 21. Review.
|







