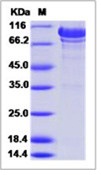
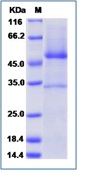
AMBP Recombinant Protein
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
| Amount : | 10 µg |
| Purification : | Greater than 90% as determined by SDS-PAGE. |
| Content : | The AMBP solution (0.5mg/ml) contains 20mM Tris-HCl buffer (pH 8.0), 1mM DTT, 0.1M NaCl and 10% glycerol. |
| Storage condition : | Store at 4°C if entire vial will be used within 2-4 weeks. Store, frozen at -20°C for longer periods of time. For long term storage it is recommended to add a carrier protein (0.1% HSA or BSA).Avoid multiple freeze-thaw cycles. |
| AA sequence : | MGSSHHHHHH SSGLVPRGSH MGPVPTPPDN IQVQENFNIS RIYGKWYNLA IGSTCPWLKK IMDRMTVSTL VLGEGATEAE ISMTSTRWRK GVCEETSGAY EKTDTDGKFL YHKSKWNITM ESYVVHTNYD EYAIFLTKKF SRHHGPTITA KLYGRAPQLR ETLLQDFRVV AQGVGIPEDS IFTMADRGEC VPGEQEPEPI LIPRV. |
| Alternative Name : | Alpha-1-microglobulin/bikunin precursor, HCP, ITIL, ITI, EDC1, HI30, IATIL, ITILC, UTI, A1M, bikunin, complex-forming glycoprotein heterogeneous in charge, growth-inhibiting protein 19, inter-alpha-trypsin inhibitor light chain, protein AMBP |
Source : E.coli. AMBP Human Recombinant produced in E.coli is a single, non-glycosylated polypeptide chain containing 205 amino acids (20-203) and having a molecular mass of 23.1 kDa.AMBP is fused to a 21 amino acid His-tag at N-terminus & purified by proprietary chromatographic techniques. Alpha 1-microglobulin (A1M) is a lipocalin superfamily member (kernal lipocalins). A1M is a low molecular weight protein component of plasma. A1M is distributed in plasma and extravascular compartments of all organs. Alpha-1 Microglobulin is found in mammals, birds, amphibians and fish. The primary sites of A1M synthesis are the liver and the kidney. Around the opening of the lipocalin pocket three lysyl residues are situated; those residues carry yellow-brown modification derived from the binding and degradation of heme and kynurenin (a tryptophan metabolite). A1-Microglobulin's reductase and dehydrogenase have broad biological substrate specificity properties due to its' free cysteine side-chain which is located in a flexible loop. Alpha-1-microglobulin is glycosylated by three separate carbohydrate chains: two complex carbohydrates which are N-linked to asparagines at residues 17 and 96, and the other simple carbohydrate which is O-linked to threonine at position 5. The carbohydrates comprise 22% of the total molecular mass of the protein. The glycosylation varies between species. A1M exists in two forms- a free form and complexed to other macromolecules: in humans- complexed to immunoglobulin A (IgA), in rat- complexed to alpha-1-inhibitor-3. Free A1M is exceptionally heterogeneous in charge (therefore also known as protein HC), and is found tightly linked to a chromophore. The free Alpha-1-microglobulin is a monomeric protein composed of one 188 residue polypeptide and contains three cysteines, two of which (residues 75 and 173) form a conserved intra-molecular disulphide link. The chromophoric group is covalently bound to the free cysteine residue at position 34. A1M binds retinol as a major ligand, but this is probably distinct from its covalent chromophore. Half of all human plasma A1M (approximately 0.03mg/ml) forms a 1:1 complex with about 5% of plasma immunoglobulin A. The resulting macromolecular complexes' molecular weight is 200000, and a plasma concentration of 0.3mg/ml. The complex can exhibit both antibody activity and affect many of the biological actions of free Alpha-1-microglobulin. Alpha-1-microglobulin was first discovered in pathological human urine.It was suggested that A1M might be involved in tissue defense against reactive oxygen species, oxidation by heme and kynurenin. Evidence also suggests that A1M functions in the regulation of the immune system. Other functions include inhibition of stimulation of cultured lymphocytes by protein antigens; induction of cell division of lymphocytes, a mitogenic effect that can either be enhanced or inhibited by the action of other plasma components; inhibition of neutrophil granulocyte migration in vitro; and inhibition of chemotaxis.
|
There are currently no product reviews
|











.png)