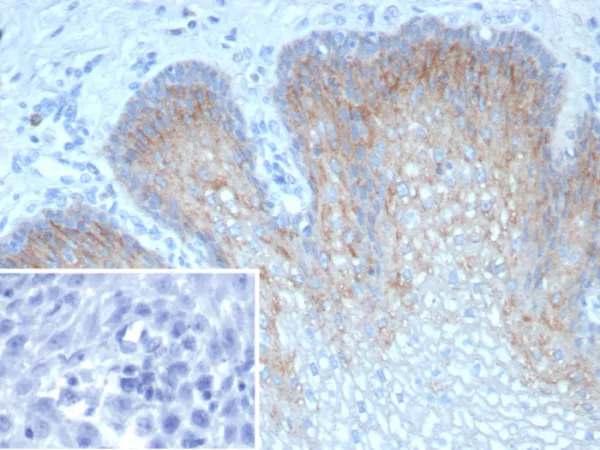
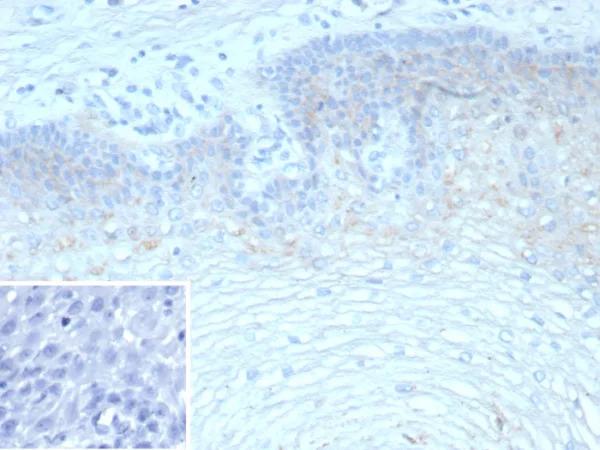
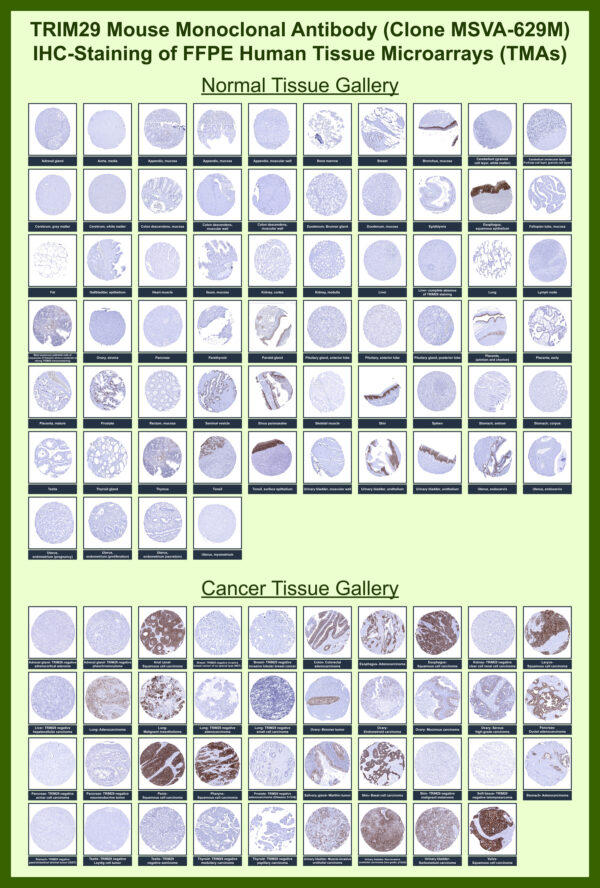
Clusterin Mouse
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
| Amount : | 10 µg |
| Purification : | Greater than 90.0% as determined by SDS-PAGE. |
| Content : | Clusterin filtered (0.4 µm) and lyophilized from 0.5mg/ml in 20 mM Tris buffer and 50 mM NaCl, pH 7.5. It is recommended to add deionized water to prepare a working stock solution of approximately 0.5mg/ml and let the lyophilized pellet dissolve completely. |
| Storage condition : | Store lyophilized protein at -20°C. Aliquot the product after reconstitution to avoid repeated freezing/thawing cycles. Reconstituted protein can be stored at 4°C for a limited period of time; it does not show any change after two weeks at 4°C. |
| AA sequence : | EQEVSDNELQ ELSTQGSRYI NKEIQNAVQG VKHIKTLIEK TNAERKSLLN SLEEAKKKKE DALEDTRDSE MKLKAFPEVC NETMMALWEE CKPCLKHTCM KFYARVCRSG SGLVGQQLEE FLNQSSPFYF WMNGDRIDSL LESDRQQSQV LDAMQDSFAR ASGIIDTLFQ DRFFARELHD PHYFSPIGFP HKRPHFLYPK SRLVRSLMSP SHYGPPSFHN MFQPFFEMIH QAQQAMDVQL HSPAFQFPDV DFLREGEDDR TVCKEIRRNS TGCLKMKGQC EKCQEILSVD CSTNNPAQAN LRQELNDSLQ VAERLTEQYK ELLQSFQSKM LNTSSLLEQL NDQFNWVSQL ANLTQGEDKY YLRVSTVTTH SSDSEVPSRV TEVVVKLFDS DPITVVLPEE VSKDNPKFMD TVAEKALQEY RRKSRAEHHH HHH |
| Alternative Name : | CLI, AAG4, KUB1, SGP2, SGP-2, SP-40, TRPM2, MGC24903, Clusterin, Apolipoprotein J, Apo-J. |
Source: HEK293
Filtered White lyophilized (freeze-dried) powder.
Clusterin also known as Apolipoprotein J (APO-J) is a 75-80 kD disulfide-linked heterodimeric protein containing about 30% of N-linked carbohydrate rich in sialic acid but truncated forms targeted to the nucleus have also been identified. The precursor polypeptide chain is cleaved proteolytically to remove the 22-mer secretory signal peptide and subsequently between residues 227/228 to generate the a and b chains. These are assembled in anti-parallel to give a heterodimeric molecule in which the cysteine-rich centers are linked by 5 disulfide bridges and are flanked by 2 predicted coiled-coil a-helices and 3 predicted amphipathic a-helices. Across a broad range of species clusterin shows a high degree of sequence homology ranging from 70% -80%. It is nearly ubiquitously expressed in most mammalian tissues and can be found in plasma, milk, urine, cerebrospinal fluid and semen. It is able to bind and form complexes with numerous partners such as immunoglobulins, lipids, heparin, bacteria, complement components, paraoxonase, beta amyloid, leptin and others. Clusterin has been ascribed a plethora of functions such as phagocyte recruitment, aggregation induction, complement attack prevention, apoptosis inhibition, membrane remodeling, lipid transport, hormone transport and/or scavenging, matrix metalloproteinase inhibition. Clusterin is up/down regulated on the mRNA or protein level in many pathological and clinically relevant situations including cancer, organ regeneration, infection, Alzheimer disease, retinitis pigmentosa, myocardial infarction, renal tubular damage, autoimmunity and others.
Clusterin Mouse Recombinant is a single, glycosylated polypeptide chain containing 433 amino acids (22-448a.a) and having a molecular mass of 50.2kDa (calculated). Clusterin is fused to a 6 a.a His tag at C-terminal.
Filtered White lyophilized (freeze-dried) powder.
Clusterin also known as Apolipoprotein J (APO-J) is a 75-80 kD disulfide-linked heterodimeric protein containing about 30% of N-linked carbohydrate rich in sialic acid but truncated forms targeted to the nucleus have also been identified. The precursor polypeptide chain is cleaved proteolytically to remove the 22-mer secretory signal peptide and subsequently between residues 227/228 to generate the a and b chains. These are assembled in anti-parallel to give a heterodimeric molecule in which the cysteine-rich centers are linked by 5 disulfide bridges and are flanked by 2 predicted coiled-coil a-helices and 3 predicted amphipathic a-helices. Across a broad range of species clusterin shows a high degree of sequence homology ranging from 70% -80%. It is nearly ubiquitously expressed in most mammalian tissues and can be found in plasma, milk, urine, cerebrospinal fluid and semen. It is able to bind and form complexes with numerous partners such as immunoglobulins, lipids, heparin, bacteria, complement components, paraoxonase, beta amyloid, leptin and others. Clusterin has been ascribed a plethora of functions such as phagocyte recruitment, aggregation induction, complement attack prevention, apoptosis inhibition, membrane remodeling, lipid transport, hormone transport and/or scavenging, matrix metalloproteinase inhibition. Clusterin is up/down regulated on the mRNA or protein level in many pathological and clinically relevant situations including cancer, organ regeneration, infection, Alzheimer disease, retinitis pigmentosa, myocardial infarction, renal tubular damage, autoimmunity and others.
Clusterin Mouse Recombinant is a single, glycosylated polypeptide chain containing 433 amino acids (22-448a.a) and having a molecular mass of 50.2kDa (calculated). Clusterin is fused to a 6 a.a His tag at C-terminal.
|
There are currently no product reviews
|







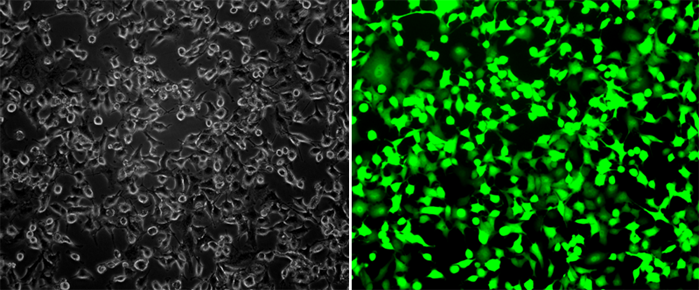






.png)