FTL1 Human, HEK(Discontinued)
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
| Amount : | 10 µg |
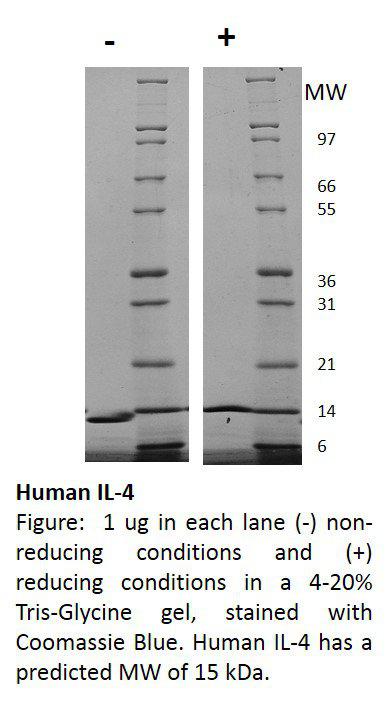
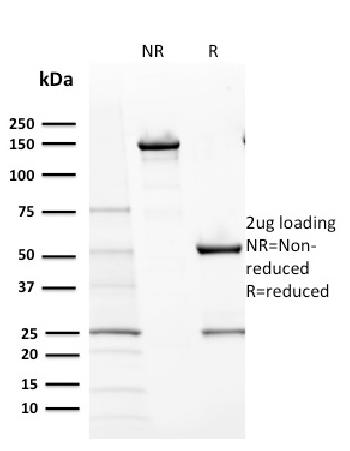
| Purification : | Greater than 95.0% as determined by SDS-PAGE. |
| Content : | FTL1 protein solution is filtered in in PBS pH 7.4 and 20% (w/v) glycerol. |
| Storage condition : | Store at 4°C if entire vial will be used within 2-4 weeks.Store, frozen at -20°C for longer periods of time.Avoid multiple freeze-thaw cycles. |
| AA sequence : | ASSKLKDPEL SLKGTQHIMQ AGQTLHLQCR GEAAHKWSLP EMVSKESERL SITKSACGRN GKQFCSTLTL NTAQANHTGF YSCKYLAVPT SKKKETESAI YIFISDTGRP FVEMYSEIPE IIHMTEGREL VIPCRVTSPN ITVTLKKFPL DTLIPDGKRI IWDSRKGFII SNATYKEIGL LTCEATVNGH LYKTNYLTHR QTNTIIDVQI STPRPVKLLR GHTLVLNCTA TTPLNTRVQM TWSYPDEKNK RASVRRRIDQ SNSHANIFYS VLTIDKMQNK DKGLYTCRVR SGPSFKSVNT SVHIYDKAFI TVKHRKQQVL ETVAGKRSYR LSMKVKAFPS PEVVWLKDGL PATEKSARYL TRGYSLIIKD VTEEDAGNYT ILLSIKQSNV FKNLTATLIV NVKPQIYEKA VSSFPDPALY PLGSRQILTC TAYGIPQPTI KWFWHPCNHN HSEARCDFCS NNEESFILDA DSNMGNRIES ITQRMAIIEG KNKMASTLVV ADSRISGIYI CIASNKVGTV GRNISFYITD VPNGFHVNLE KMPTEGEDLK LSCTVNKFLY RDVTWILLRT VNNRTMHYSI SKQKMAITKE HSITLNLTIM NVSLQDSGTY ACRARNVYTG EEILQKKEIT IRGEHCNKKA VFSRISKFKS TRNDCTTQSN VKHKLHHHHH HHHHH |
| Alternative Name : | FLT-1, FLT1, Tyrosine-protein kinase receptor FLT, Flt-1, Tyrosine-protein kinase FRT, Fms-like tyrosine kinase 1, VEGFR-1. |
Source: HEK293
Filtered clear solution.
Endothelial cells express 3 different vascular endothelial growth factor (VEGF) receptors, belonging to the family of receptor tyrosine kinases (RTKs). They are named VEGFR-1 (Flt-1), VEGFR-2 (KDR/Flk-1), VEGFR-3 (Flt-4). Their expression is almost exclusively restricted to endothelial cells, but VEGFR-1 can also be found on monocytes, dendritic cells and on trophoblast cells. The flt-1 gene was first described in 1990. The receptor contains seven immunoglobulin-like extracellular domains, a single transmembrane region and an intracellular splited tyrosine kinase domain. Compared to VEGFR-2 the Flt-1 receptor has a higher affinity for VEGF but a weaker signaling activity. VEGFR-1 thus leads not to proliferation of endothelial cells, but mediates signals for differentiation. Interestingly a naturally occuring soluble variant of VEGFR-1 (sVEGFR-1) was found in HUVE supernatants in 1996, which is generated by alternative splicing of the flt-1 mRNA. The biological functions of sVEGFR-1 still are not clear, but it seems to be an endogenous regulator of angiogenesis, binding VEGF with the same affinity as the full-length receptor.
FTL1 Human Recombinant is a single, glycosylated polypeptide chain containing 675 amino acids (27-687a.a) and having a molecular mass of 76.6kDa (calculated). FTL1 is composed of a 2 a.a N-terminal linker, a 2 a.a C-terminal linker and fused to a 10 a.a His tag at C-Terminus.
Filtered clear solution.
Endothelial cells express 3 different vascular endothelial growth factor (VEGF) receptors, belonging to the family of receptor tyrosine kinases (RTKs). They are named VEGFR-1 (Flt-1), VEGFR-2 (KDR/Flk-1), VEGFR-3 (Flt-4). Their expression is almost exclusively restricted to endothelial cells, but VEGFR-1 can also be found on monocytes, dendritic cells and on trophoblast cells. The flt-1 gene was first described in 1990. The receptor contains seven immunoglobulin-like extracellular domains, a single transmembrane region and an intracellular splited tyrosine kinase domain. Compared to VEGFR-2 the Flt-1 receptor has a higher affinity for VEGF but a weaker signaling activity. VEGFR-1 thus leads not to proliferation of endothelial cells, but mediates signals for differentiation. Interestingly a naturally occuring soluble variant of VEGFR-1 (sVEGFR-1) was found in HUVE supernatants in 1996, which is generated by alternative splicing of the flt-1 mRNA. The biological functions of sVEGFR-1 still are not clear, but it seems to be an endogenous regulator of angiogenesis, binding VEGF with the same affinity as the full-length receptor.
FTL1 Human Recombinant is a single, glycosylated polypeptide chain containing 675 amino acids (27-687a.a) and having a molecular mass of 76.6kDa (calculated). FTL1 is composed of a 2 a.a N-terminal linker, a 2 a.a C-terminal linker and fused to a 10 a.a His tag at C-Terminus.
|
There are currently no product reviews
|













.png)








