Polyclonal Antibody to GAPDH

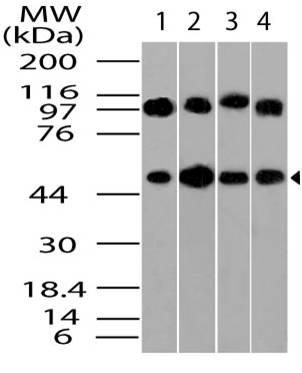
Figure-1: Western blot analysis of GAPDH. Anti- GAPDH antibody (11-13024) was used at 1 µg/ml on (1) h Brain, (2) h Heart, (3) h Small Intestine, (4) h Kidney, (5) h Liver, (6) h Lung, (7) h Skeletal Muscle, (8) h Stomach, (9) h Spleen, (10) h Ovary, (11) h Testis lysates.
Roll over image to zoom in
Shipping Info:
Order now and get it on Tuesday March 17, 2026
Same day delivery FREE on San Diego area orders placed by 1.00 PM
| Format : | Purified |
| Amount : | 100 µg |
| Isotype : | Rabbit IgG |
| Purification : | Protein A Chromatography |
| Content : | 25 µg in 50 µl/100 µg in 200 µl PBS containing 0.05% BSA and 0.05% sodium azide. Sodium azide is highly toxic. |
| Storage condition : | Store the antibody at 4°C, stable for 6 months. For long-term storage, store at -20°C. Avoid repeated freeze and thaw cycles. |
GAPDH (Glyceraldehyde-3-Phosphate Dehydrogenase) is an enzyme best known for its role in glycolysis. However, extra-glycolytic functions of GAPDH have been described, including regulation of protein expression via RNA binding. GAPDH binds to numerous AREs (adenine-uridine rich elements) from various mRNA 3'-untranslated regions in vitro and in vivo despite its lack of a canonical RNA binding motif. GAPDH specifically catalyzes the simultaneous phosphorylation and oxidation of glyceraldehyde 3-phosphate using NAD+ (Nicotinamide Adenine Dinucleotide) as a cofactor to produce glycerate 1,3-biphosphate and NADH. In addition to its role in energy production, GAPDH has been implicated in many cellular processes including DNA repair tRNA export, membrane fusion and transport, endocytosis and nuclear membrane assembly, and cell death.
Western blot analysis: 1-2 µg/ml
For Research Use Only. Not for use in diagnostic/therapeutics procedures.
| Subcellular location: | Cytoplasm, Nucleus, Cytoplasm, Membrane, Cytoplasm |
| Post transnational modification: | Oxidative stress can promote the formation of high molecular weight disulfide-linked GAPDH aggregates, through a process called nucleocytoplasmic coagulation. Such aggregates can be observed in vivo in the affected tissues of patients with Alzheimer disease or alcoholic liver cirrhosis, or in cell cultures during necrosis. Oxidation at Met-46 may play a pivotal role in the formation of these insoluble structures. This modification has been detected in vitro following treatment with free radical donor (+/-)-(E)-4-ethyl-2-[(E)-hydroxyimino]-5-nitro-3-hexenamide. It has been proposed to destabilize nearby residues, increasing the likelihood of secondary oxidative damages, including oxidation of Tyr-45 and Met-105. This cascade of oxidations may augment GAPDH misfolding, leading to intermolecular disulfide cross-linking and aggregation. |
| BioGrid: | 108868. 233 interactions. |
|
There are currently no product reviews
|









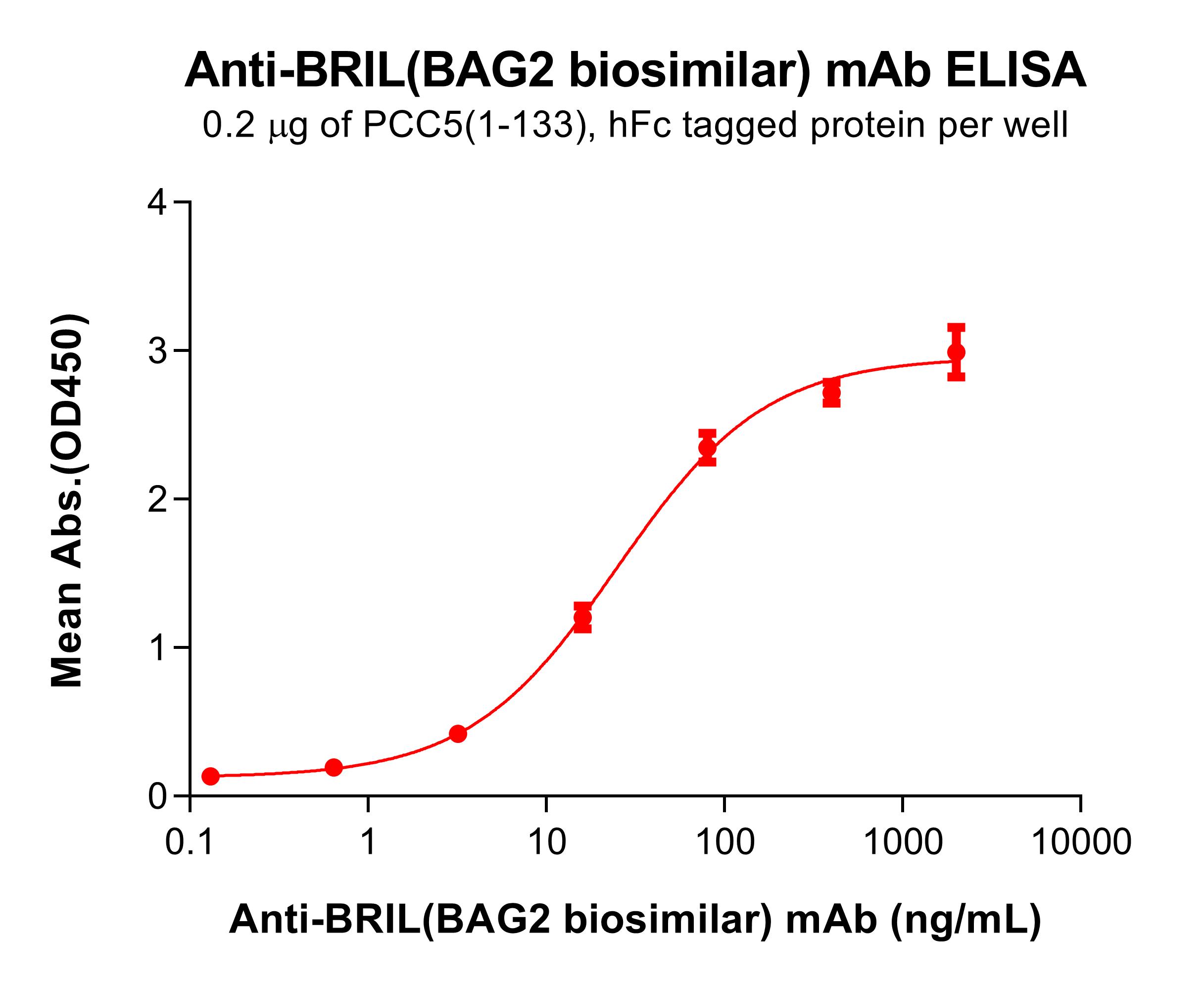
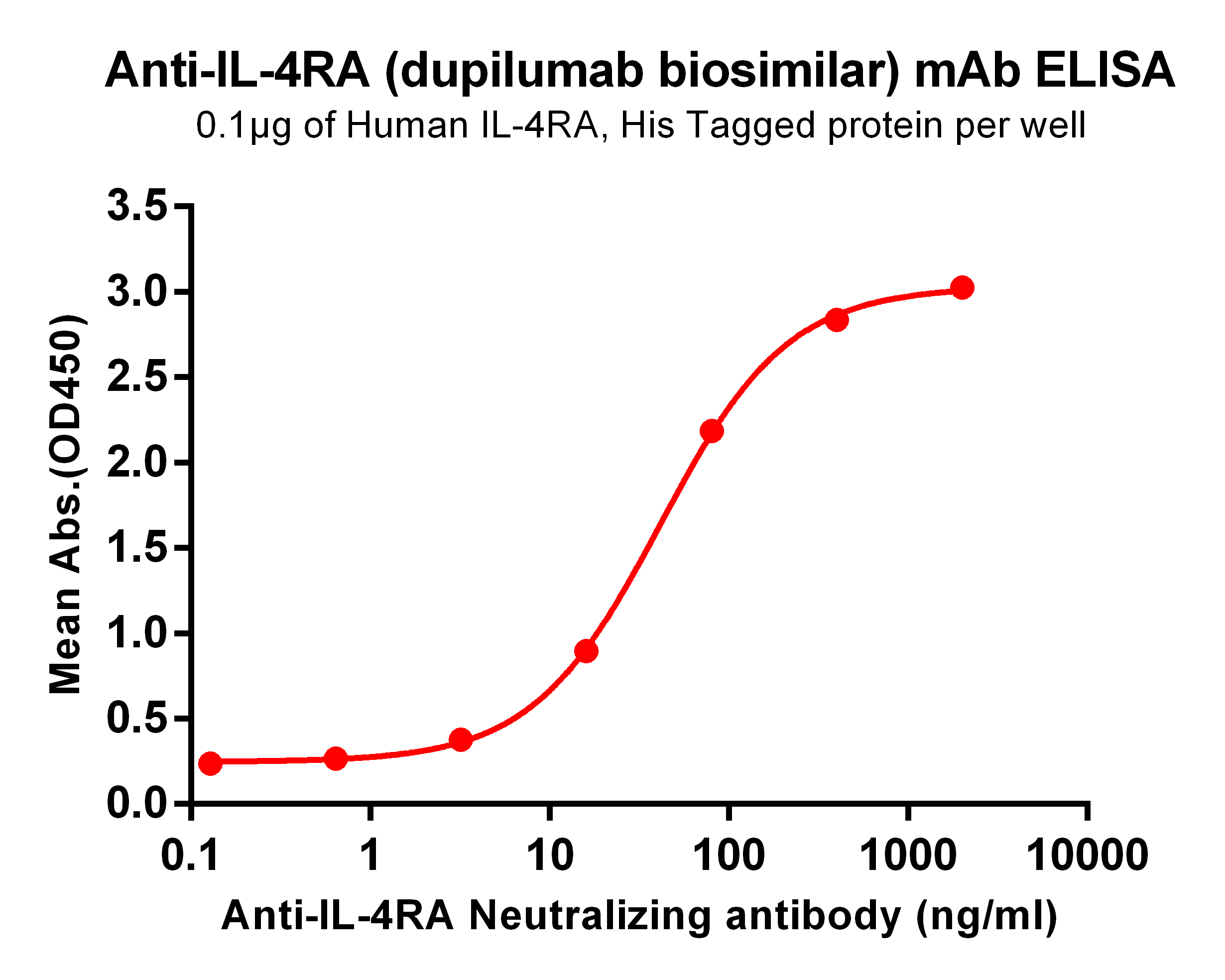






.png)