Recombinant Aeromonas Aminopeptidase(Discontinued)
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
| Amount : | 500 µg |
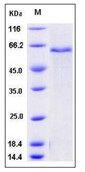
| Purification : | Purity: >= 98% by SDS-PAGE gel and HPLC analyses. |
| AA sequence : | MPPITQQATV TAWLPQVDAS QITGTISSLE SFTNRFYTTT SGAQASDWIA SEWQALSASL PNASVKQVSH SGYNQKSVVM TITGSEAPDE WIVIGGHLDS TIGSHTNEQS VAPGADDDASGIAAVTEVIR VLSENNFQPK RSIAFMAYAA EEVGLRGSQD LANQYKSEGK NVVSALQLDM TNYKGSAQDV VFITDYTDSN FTQYLTQLMD EYLPSLTYGF DTCGYACSDH ASWHNAGYPAAMPFESKFND YNPRIHTTQD TLANSDPTGS HAKKFTQLGL AYAIEMGSAT G |
Source: E.coli
Proteases (also called Proteolytic Enzymes, Peptidases, or Proteinases) are enzymes that hydrolyze the amide bonds within proteins or peptides. Most proteases act in a specific manner, hydrolyzing bonds at, or adjacent to specific residues, or a specific sequence of residues contained within the substrate protein or peptide. Proteases play an important role in most diseases and biological processes, including prenatal and postnatal development, reproduction, signal transduction, the immune response, various autoimmune and degenerative diseases, and cancer. They are also an important research tool, frequently used in the analysis and production of proteins. Recombinant Aeromonas Aminopeptidase is a 31.4 kDa protein containing 291 amino acid residues.
Sequentially cleaves N-terminal amino acids except E, D, and X-P.
For Research Use Only. Not for use in diagnostic/therapeutics procedures.
|
There are currently no product reviews
|













.png)









