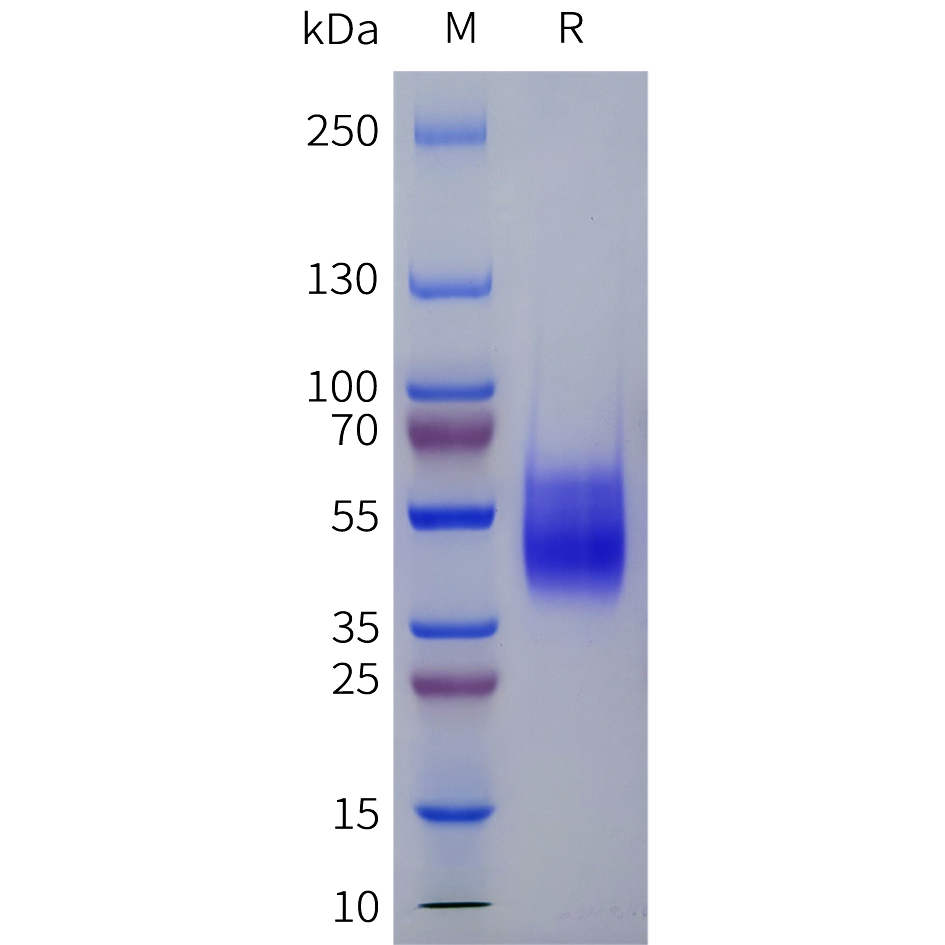
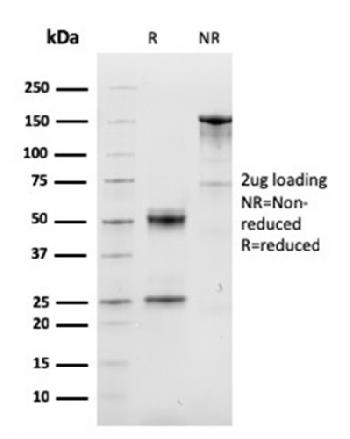
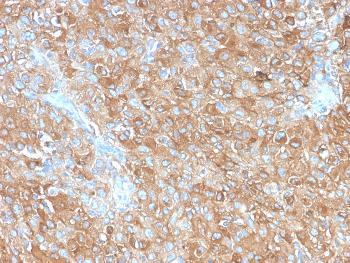
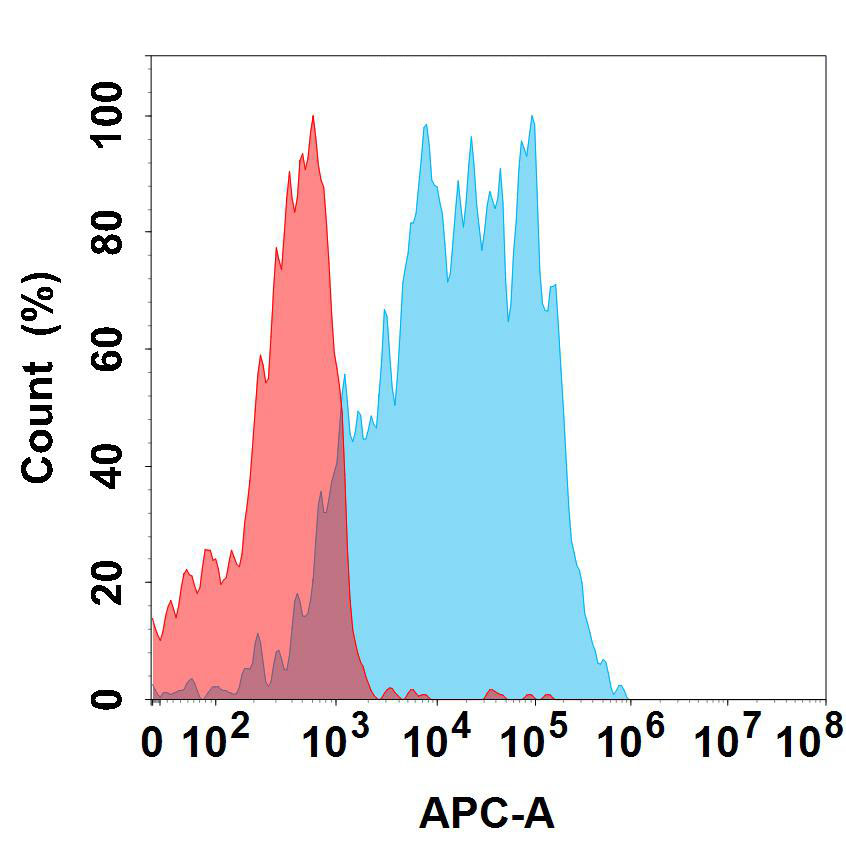
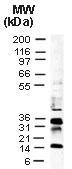
Recombinant Human IDO-2/INDOL1(C-6His)
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
| Amount : | 50 µg |
| Content : | Supplied as a 0.2 µm filtered solution of 20mM Tris, 10%Glycerol, 1mM EDTA, 250mM NaCl,pH8.0. |
| AA sequence : | Recombinant Human Indoleamine 2,3-dioxygenase 2 is produced by our E.coli expression system and the target gene encoding Met14-Gly420 is expressed with a 6His tag at the C-terminus. |
| Alternative Name : | Indoleamine 2,3-dioxygenase 2;Indoleamine 2,3-dioxygenase-like protein 1;Indoleamine-pyrrole 2,3-dioxygenase-like protein 1;IDO2;INDOL1 |
Source : E. coli;
Indoleamine 2,3-dioxygenase-like protein 1(IDO2) belongs to the indoleamine 2,3-dioxygenase family. IDO2 can be detected in liver, small intestine, spleen, placenta, thymus, lung, brain, kidney, and colon. It also expressed at low level in testis and thyroid but not expressed in the majority of human tumor samples. IDO2 catalyzes the first and rate limiting step of the catabolism of the essential amino acid tryptophan along the kynurenine pathway. It involved in immune regulation. IDO1 and IDO2 are 2 distinct enzymes which catalyze the same reaction. IDO2 affinity for tryptophan is much lower than that of IDO1. 50 % of Caucasians harbor polymorphisms which abolish IDO2 enzymatic activity. IDO2 is expressed in human tumors in an inactive form: tryptophan degradation is entirely provided by IDO1 in these cells. IDO2 may play a role as a negative regulator of IDO1 by competing for heme-binding with IDO1. Low efficiency IDO2 enzymes have been conserved throughout vertebrate evolution, whereas higher efficiency IDO1 enzymes are dispensable in many lower vertebrate lineages. IDO1 may have arisen by gene duplication of a more ancient proto-IDO gene before the divergence of marsupial and eutherian (placental) mammals.
Indoleamine 2,3-dioxygenase-like protein 1(IDO2) belongs to the indoleamine 2,3-dioxygenase family. IDO2 can be detected in liver, small intestine, spleen, placenta, thymus, lung, brain, kidney, and colon. It also expressed at low level in testis and thyroid but not expressed in the majority of human tumor samples. IDO2 catalyzes the first and rate limiting step of the catabolism of the essential amino acid tryptophan along the kynurenine pathway. It involved in immune regulation. IDO1 and IDO2 are 2 distinct enzymes which catalyze the same reaction. IDO2 affinity for tryptophan is much lower than that of IDO1. 50 % of Caucasians harbor polymorphisms which abolish IDO2 enzymatic activity. IDO2 is expressed in human tumors in an inactive form: tryptophan degradation is entirely provided by IDO1 in these cells. IDO2 may play a role as a negative regulator of IDO1 by competing for heme-binding with IDO1. Low efficiency IDO2 enzymes have been conserved throughout vertebrate evolution, whereas higher efficiency IDO1 enzymes are dispensable in many lower vertebrate lineages. IDO1 may have arisen by gene duplication of a more ancient proto-IDO gene before the divergence of marsupial and eutherian (placental) mammals.
|
There are currently no product reviews
|













.png)