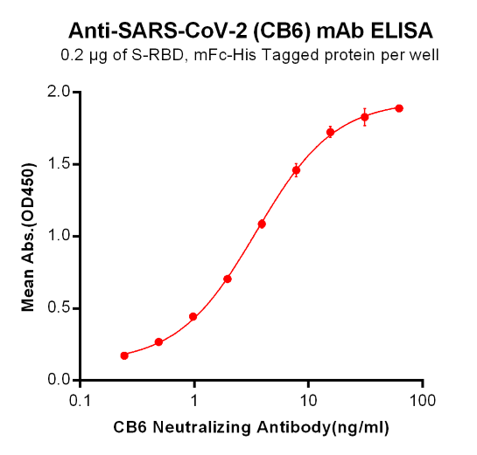
Anti-Human IL 12/23 (Briakinumab)
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
| Amount : | 100 µg |
| Isotype : | Human IgG1lambda |
| Purification : | >=95% monomer by analytical SEC |
| Content : | Concentration : >= 5.0 mg/ml This biosimilar antibody is aseptically packaged and formulated in 0.01 M phosphate buffered saline (150 mM NaCl) PBS pH 7.2 - 7.4 with no carrier protein, potassium, calcium or preservatives added. |
| Storage condition : | Functional grade biosimilar antibodies may be stored sterile as received at 2-8°C for up to one month. For longer term storage, aseptically aliquot in working volumes without diluting and store at -80°C. Avoid Repeated Freeze Thaw Cycles. |
| Alternative Name : | IL-12p40; Interleukin 12; Interleukin 23; IL12; IL23; IL-12; IL-23 |
| Immunogen Information : | This antibody was produced by phage display technology. |
Expression Host : HEK-293
Pathogen Testing : To protect mouse colonies from infection by pathogens and to assure that experimental preclinical data is not affected by such pathogens, all of this recombinant biosimilar antibodies are tested and guaranteed to be negative for all pathogens in the IDEXX IMPACT I Mouse Profile.
This non-therapeutic biosimilar antibody uses the same variable region sequence as the therapeutic antibody Briakinumab. Briakinumab recognizes both human IL12 and IL23 via IL-12/23p40. This product is for research use only.
Briakinumab is a human monoclonal antibody targets the p40 subunit shared by interleukins 12 and 23. IL-12 associates with IL-23alpha to form the heterodimeric cytokine IL-23. IL-23 is associated with various autoimmune inflammatory diseases, and is particularly highly expressed in psoriasis skin lesions. In addition, IL-23 is suspected to play a role in tumorigenesis. Briakinumab binds to and neutralizes human IL-12 and IL-23 (via their shared p40 subunit) and is being investigated for the treatment of rheumatoid arthritis, inflammatory bowel disease, and multiple sclerosis. Anti-Human IL 12/23 (Briakinumab) utilizes the same variable regions from the therapeutic antibody Briakinumab making it ideal for research projects.
Endotoxin : <= 1.0 EU/mg as determined by the LAL method
The suggested concentration for Briakinumab biosimilar antibody for staining cells in flow cytometry is <= 0.25 µg per 106 cells in a volume of 100 µl. Titration of the reagent is recommended for optimal performance for each application.
For Research Use Only. Not for use in diagnostic/therapeutics procedures.
|
There are currently no product reviews
|














.png)