Monoclonal Antibody to Ebola GP II (Clone: ABM4C78)

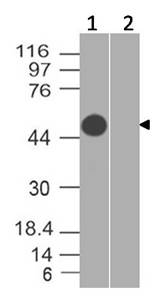
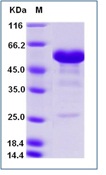
Fig-1: Western blot analysis of Ebola GP II. Anti-Ebola GP II antibody (Clone: ABM4C78) was tested at 0.1 µg/ml partial length recombinant protein.
Roll over image to zoom in
Shipping Info:
Order now and get it on Thursday June 04, 2026
Same day delivery FREE on San Diego area orders placed by 1.00 PM
| Format : | Purified |
| Amount : | 100 µg |
| Isotype : | Mouse IgG2a Lambda |
| Purification : | Protein G Chromatography |
| Content : | 25 µg in 50 µl/100 µg in 200 µl PBS containing 0.05% BSA and 0.05% sodium azide. Sodium azide is highly toxic. |
| Storage condition : | Store the antibody at 4°C; stable for 6 months. For long-term storage; store at -20°C. Avoid repeated freeze and thaw cycles. |
The Sudan ebola virus (SUDV) glycoprotein (GP) is an envelope glycoprotein that is present on the virion surface and is involved in receptor binding and mediating viral entry. It is composed of a trimer of heterodimers (GP1/GP2), where GP1 and GP2 remain covalently linked by a disulfide bond9, and the resulting GP1-GP2 pair trimerizes to form a ~450 kDa envelope spike on the viral surface. GP is synthesized as a single polypeptide of 676 amino acids in length that is post-translationally cleaved by furin to yield two subunits, GP1 and GP2. The GP2 subunit mediates virus-cell membrane fusion and viral entry. Structural analysis of the core domain of Ebola virus GP2 indicates that the fusion-active conformation of GP2 is similar to that of the other class I membrane fusion proteins, suggesting a common membrane fusion mechanism. In the endosome, GP2 releases from GP1 and undergoes irreversible conformational changes that drive fusion with host endosomal membranes. GP2 contains an N-terminal peptide, a hairpin-forming fusion loop, and two heptad repeats connected by a functionally important linker. The first heptad repeat of GP2 is wound around the base of GP1 in a metastable, prefusion-specific conformation.
Western blot analysis: 0.5-1 µg/ml
For Research Use Only. Not for use in diagnostic/therapeutics procedures.
| Subcellular location: | Secreted |
| Post transnational modification: | Specific enzymatic cleavages in vivo yield mature proteins. The precursor is processed into GP1 and GP2 by host cell furin in the trans Golgi, and maybe by other host proteases, to yield the mature GP1 and GP2 proteins. The cleavage site corresponds to the furin optimal cleavage sequence [KR]-X-[KR]-R. This cleavage does not seem to be required for function. After the internalization of the virus into cell endosomes, GP1 C-terminus is removed by the endosomal proteases cathepsin B, cathepsin L, or both, leaving a 19-kDa N-terminal fragment which is further digested by cathepsin B. Proteolytic processing of GP1,2 by host ADAM17 can remove the transmembrane anchor of GP2 and leads to shedding of complexes consisting in GP1 and truncated GP2 (GP1,2delta) (By similarity). |
|
There are currently no product reviews
|












.png)