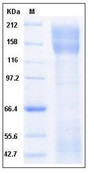
Recombinant human ANPEP protein with C-terminal human Fc tag
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
| Amount : | 50 µg |
| Purification : | The purity of the protein is greater than 95% as determined by SDS-PAGE and Coomassie blue staining. |
| Content : | Lyophilized from sterile PBS, pH 7.4. Normally 5 % - 8 % trehalose is added as protectants before lyophilization. |
| Storage condition : | Store at -80°C for 12 months (Avoid repeated freezing and thawing) |
| Alternative Name : | APN; CD13; GP150; LAP1; P150; PEPN |
Expression Host : HEK293
The protein has a predicted molecular mass of 128.37 kDa after removal of the signal peptide.
Aminopeptidase N is located in the small-intestinal and renal microvillar membrane, and also in other plasma membranes. In the small intestine aminopeptidase N plays a role in the final digestion of peptides generated from hydrolysis of proteins by gastric and pancreatic proteases. Its function in proximal tubular epithelial cells and other cell types is less clear. The large extracellular carboxyterminal domain contains a pentapeptide consensus sequence characteristic of members of the zinc-binding metalloproteinase superfamily. Sequence comparisons with known enzymes of this class showed that CD13 and aminopeptidase N are identical. The latter enzyme was thought to be involved in the metabolism of regulatory peptides by diverse cell types, including small intestinal and renal tubular epithelial cells, macrophages, granulocytes, and synaptic membranes from the CNS. This membrane-bound zinc metalloprotease is known to serve as a receptor for the HCoV-229E alphacoronavirus as well as other non-human coronaviruses. This gene has also been shown to promote angiogenesis, tumor growth, and metastasis and defects in this gene are associated with various types of leukemia and lymphoma.
The protein has a predicted molecular mass of 128.37 kDa after removal of the signal peptide.
Aminopeptidase N is located in the small-intestinal and renal microvillar membrane, and also in other plasma membranes. In the small intestine aminopeptidase N plays a role in the final digestion of peptides generated from hydrolysis of proteins by gastric and pancreatic proteases. Its function in proximal tubular epithelial cells and other cell types is less clear. The large extracellular carboxyterminal domain contains a pentapeptide consensus sequence characteristic of members of the zinc-binding metalloproteinase superfamily. Sequence comparisons with known enzymes of this class showed that CD13 and aminopeptidase N are identical. The latter enzyme was thought to be involved in the metabolism of regulatory peptides by diverse cell types, including small intestinal and renal tubular epithelial cells, macrophages, granulocytes, and synaptic membranes from the CNS. This membrane-bound zinc metalloprotease is known to serve as a receptor for the HCoV-229E alphacoronavirus as well as other non-human coronaviruses. This gene has also been shown to promote angiogenesis, tumor growth, and metastasis and defects in this gene are associated with various types of leukemia and lymphoma.
|
There are currently no product reviews
|






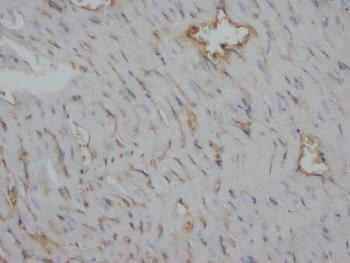
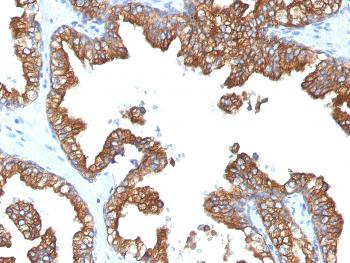
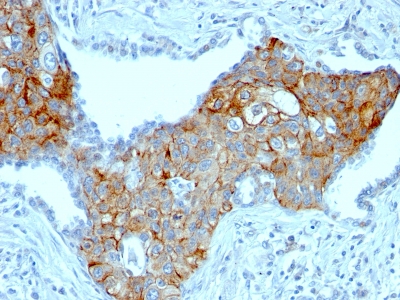
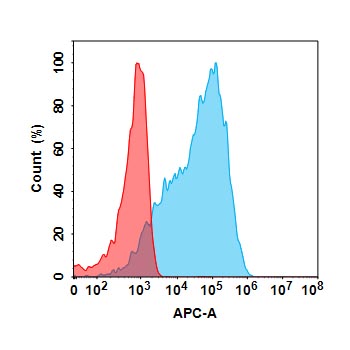







.png)