Recombinant human ZP3 protein with C-terminal human Fc tag
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
| Amount : | 50 µg |
| Purification : | The purity of the protein is greater than 95% as determined by SDS-PAGE and Coomassie blue staining. |
| Content : | Lyophilized from sterile PBS, pH 7.4. Normally 5 % - 8 % trehalose is added as protectants before lyophilization. |
| Storage condition : | Store at -80°C for 12 months (Avoid repeated freezing and thawing) |
| Alternative Name : | OOMD3; Zp-3; ZP3A; ZP3B; ZPC |
Expression Host : HEK293
The protein has a predicted molecular mass of 65.67 kDa after removal of the signal peptide.
The zona pellucida is an extracellular matrix that surrounds the oocyte and early embryo. It is composed primarily of three or four glycoproteins with various functions during fertilization and preimplantation development. The protein encoded by this gene is a structural component of the zona pellucida and functions in primary binding and induction of the sperm acrosome reaction. The nascent protein contains a N-terminal signal peptide sequence, a conserved ZP domain, a C-terminal consensus furin cleavage site, and a transmembrane domain. It is hypothesized that furin cleavage results in release of the mature protein from the plasma membrane for subsequent incorporation into the zona pellucida matrix. However, the requirement for furin cleavage in this process remains controversial based on mouse studies. A variation in the last exon of this gene has previously served as the basis for an additional ZP3 locus; however, sequence and literature review reveals that there is only one full-length ZP3 locus in the human genome. Another locus encoding a bipartite transcript designated POMZP3 contains a duplication of the last four exons of ZP3, including the above described variation, and maps closely to this gene.
The protein has a predicted molecular mass of 65.67 kDa after removal of the signal peptide.
The zona pellucida is an extracellular matrix that surrounds the oocyte and early embryo. It is composed primarily of three or four glycoproteins with various functions during fertilization and preimplantation development. The protein encoded by this gene is a structural component of the zona pellucida and functions in primary binding and induction of the sperm acrosome reaction. The nascent protein contains a N-terminal signal peptide sequence, a conserved ZP domain, a C-terminal consensus furin cleavage site, and a transmembrane domain. It is hypothesized that furin cleavage results in release of the mature protein from the plasma membrane for subsequent incorporation into the zona pellucida matrix. However, the requirement for furin cleavage in this process remains controversial based on mouse studies. A variation in the last exon of this gene has previously served as the basis for an additional ZP3 locus; however, sequence and literature review reveals that there is only one full-length ZP3 locus in the human genome. Another locus encoding a bipartite transcript designated POMZP3 contains a duplication of the last four exons of ZP3, including the above described variation, and maps closely to this gene.
|
There are currently no product reviews
|




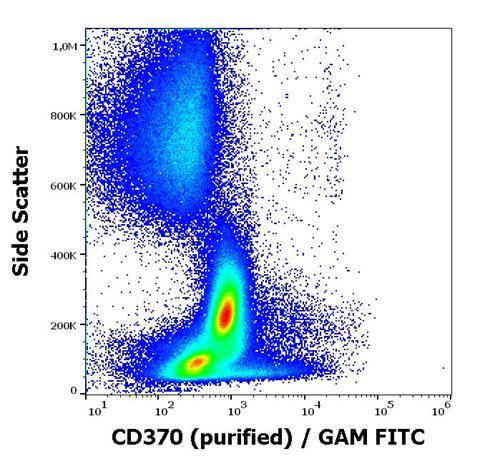


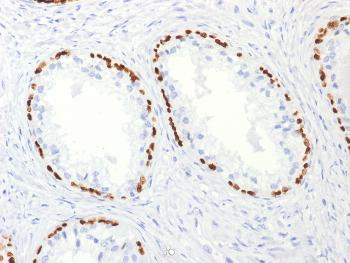







.png)








