UCHL1 Mouse, Active
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
| Amount : | 10 µg |
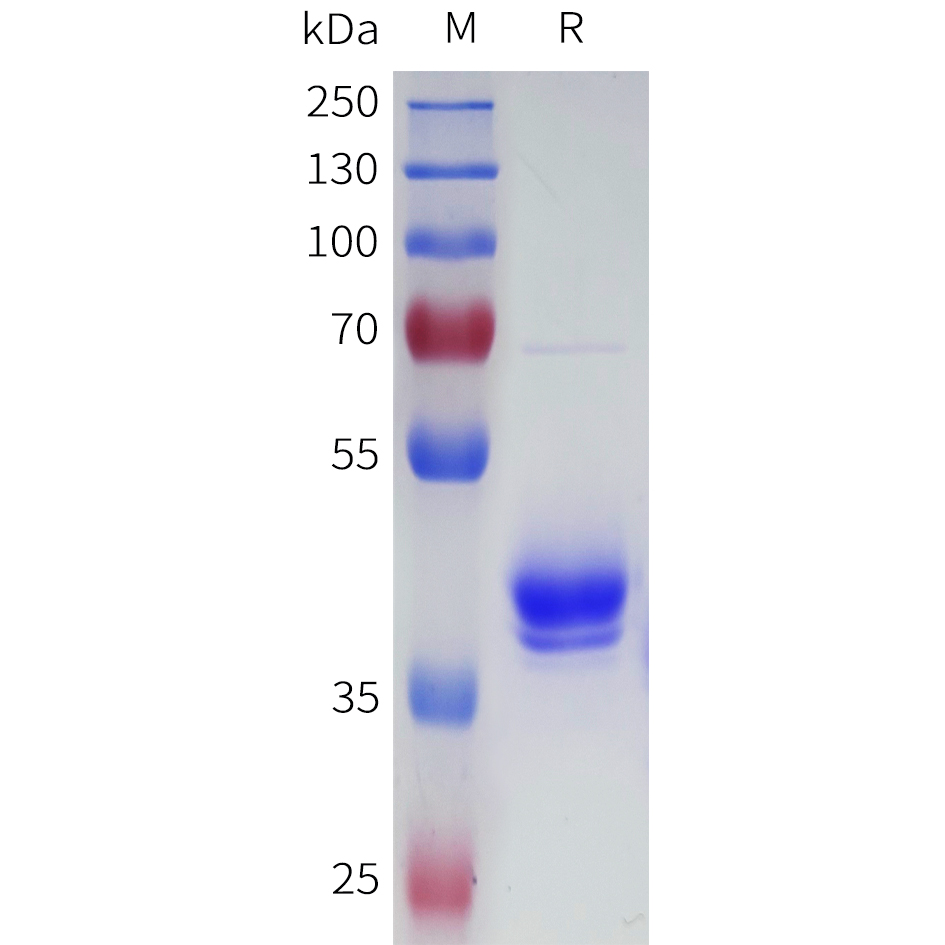
| Purification : | Greater than 90.0% as determined by SDS-PAGE. |
| Content : | UCHL1 protein solution (1mg/ml) containing Phosphate buffered saline (pH7.4), 10% glycerol and 1mM DTT. |
| Storage condition : | Store at 4°C if entire vial will be used within 2-4 weeks. Store, frozen at -20°C for longer periods of time. For long term storage it is recommended to add a carrier protein (0.1% HSA or BSA).Avoid multiple freeze-thaw cycles. |
| AA sequence : | MGSSHHHHHH SSGLVPRGSH MGSMQLKPME INPEMLNKVL AKLGVAGQWR FADVLGLEEE TLGSVPSPAC ALLLLFPLTA QHENFRKKQI EELKGQEVSP KVYFMKQTIG NSCGTIGLIH AVANNQDKLE FEDGSVLKQF LSETEKLSPE DRAKCFEKNE AIQAAHDSVA QEGQCRVDDK VNFHFILFNN VDGHLYELDG RMPFPVNHGA SSEDSLLQDA AKVCREFTER EQGEVRFSAV ALCKAA. |
| Alternative Name : | Ubiquitin carboxyl-terminal hydrolase isozyme L1, UCH-L1, Neuron cytoplasmic protein 9.5, PGP 9.5, PGP9.5, Ubiquitin thioesterase L1. |
Source: Escherichia Coli.
Sterile Filtered colorless solution.
Ubiquitin Carboxyl-Terminal Esterase L1 (UCHL1) is a part of a family whose products hydrolyze small C-terminal adducts of ubiquitin to create the ubiquitin monomer. UCHL1 is a part of the ubiquitin system, which regulates many biological activities. UCHL1 is a thiol protease that distinguishes and hydrolyzes a peptide bond at the C-terminal glycine of ubiquitin. UCHL1 binds to free monoubiquitin and avoids its degradation in lysosomes.
UCHL1 Mouse Recombinant produced in E.Coli is a single, non-glycosylated polypeptide chain containing 246 amino acids (1-223 a.a) and having a molecular mass of 27.2kDa. UCHL1 is fused to a 23 amino acid His-tag at N-terminus & purified by proprietary chromatographic techniques.
Sterile Filtered colorless solution.
Ubiquitin Carboxyl-Terminal Esterase L1 (UCHL1) is a part of a family whose products hydrolyze small C-terminal adducts of ubiquitin to create the ubiquitin monomer. UCHL1 is a part of the ubiquitin system, which regulates many biological activities. UCHL1 is a thiol protease that distinguishes and hydrolyzes a peptide bond at the C-terminal glycine of ubiquitin. UCHL1 binds to free monoubiquitin and avoids its degradation in lysosomes.
UCHL1 Mouse Recombinant produced in E.Coli is a single, non-glycosylated polypeptide chain containing 246 amino acids (1-223 a.a) and having a molecular mass of 27.2kDa. UCHL1 is fused to a 23 amino acid His-tag at N-terminus & purified by proprietary chromatographic techniques.
Specific activity is > 70 pmol/min/ug, and is defined as the amount of enzyme that hydrolysis 1.0 pmole of ubiquitin-AMC per minute at pH 7.5, at 37°C.
For Research Use Only. Not for use in diagnostic/therapeutics procedures.
|
There are currently no product reviews
|














.png)








