ctxB
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
| Amount : | 100 µg |
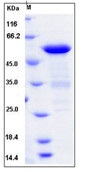
| Purification : | Greater than 98.0% as determined by:(a) Analysis by RP-HPLC.(b) Analysis by SDS-PAGE. |
| Content : | ctxB is supplied as a 0.2 µm filtered solution conteining 5mM PB, pH 7.0, 75mM NaCl, and 50 % glycerol. |
| Storage condition : | Store at 4°C if entire vial will be used within 2-4 weeks.Store, frozen at -20°C for longer periods of time.For long term storage it is recommended to add a carrier protein (0.1% HSA or BSA).Avoid multiple freeze-thaw cycles. |
| AA sequence : | TPQNITDLCA EYHNTQIYTL NDKIFSYTES LAGKREMAII TFKNGAIFQV EVPGSQHIDS QKKAIERMKD TLRIAYLTEA KVEKLCVWNN KTPHAIAAIS MAN. |
| Alternative Name : | Cholera enterotoxin subunit B, Cholera enterotoxin B chain, Cholera enterotoxin gamma chain, Choleragenoid, ctxB, toxB. |
Source: Escherichia Coli.
Sterile filtered colorless solution.
Cholera Toxin B subunit (ctxB) Cholera is a protein complex secreted by the bacterium Vibrio cholerae. ctxB is responsible for the massive, watery diarrhea characteristic of cholera infection. The cholera toxin is an oligomeric complex made up of 6 protein subunits: a single copy of the A subunit and5 copies of the B subunit, denoted as AB5. Subunit B binds while subunit A activates the G protein which activates adenylate cyclase. The five B subunits form a five-membered ring. The A subunit has 2 important segments. The A1 portion of the chain (CTA1) is a globular enzyme payload that ADP-ribosylates G proteins, while the A2 chain (CTA2) forms an extended alpha helix which sits snugly in the central pore of the B subunit ring.
Cholera Toxin B subunit Recombinant produced in E.Coli is a single, non- glycosylated polypeptide chain containing 103 amino acids and having a molecular mass of 11.6kDa.ctxB is purified by proprietary chromatographic techniques.
|
There are currently no product reviews
|













.png)








