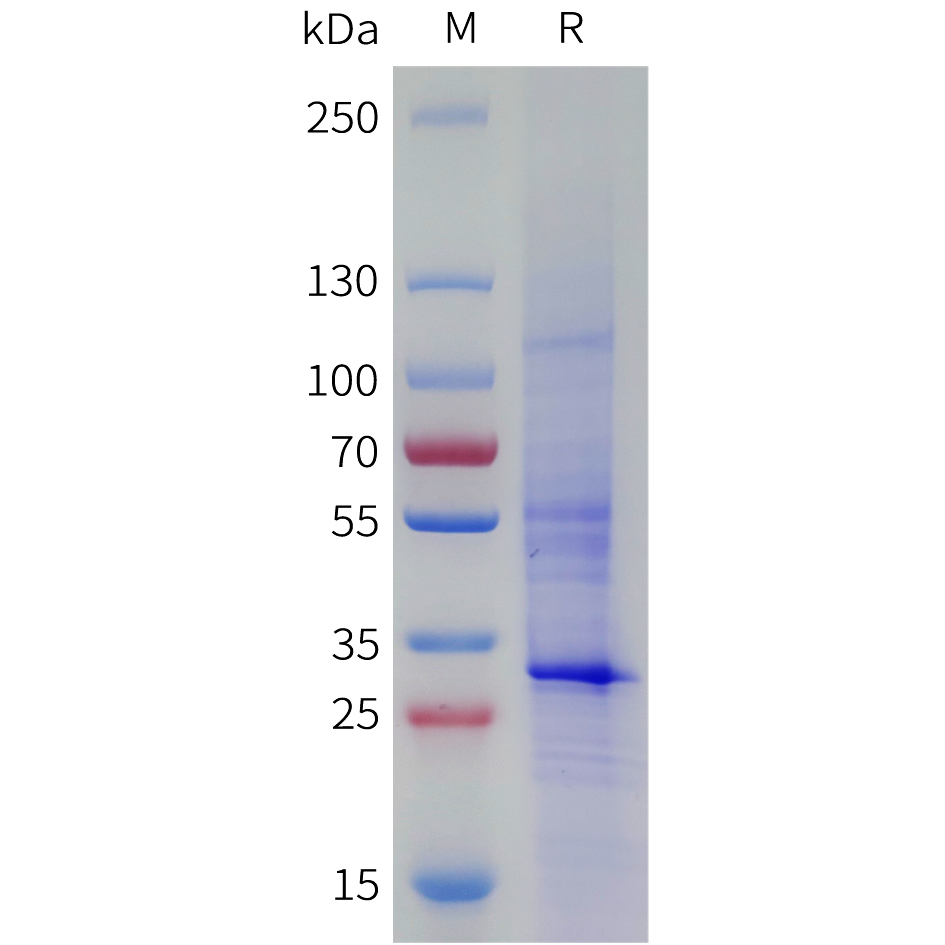
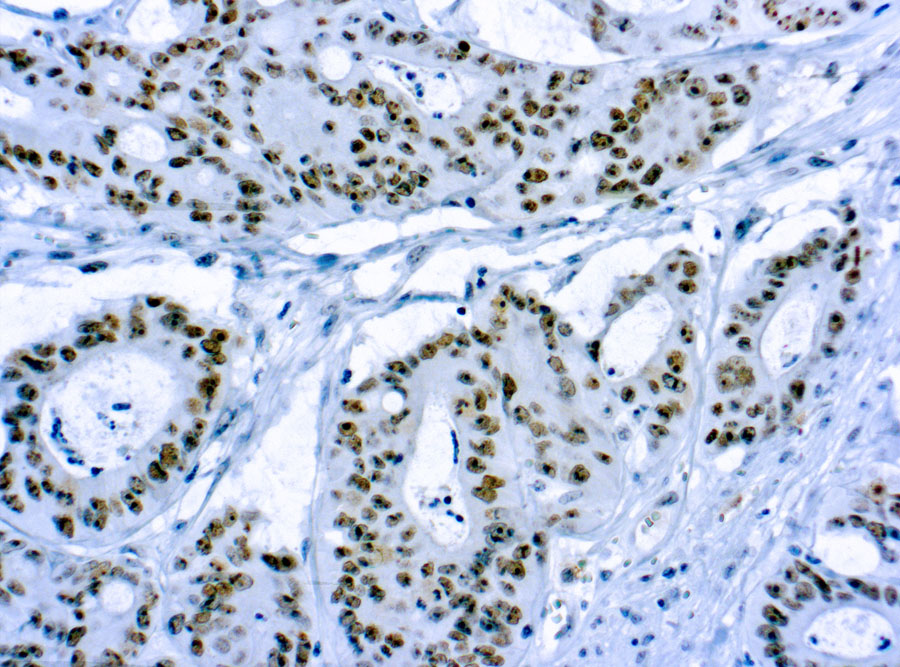
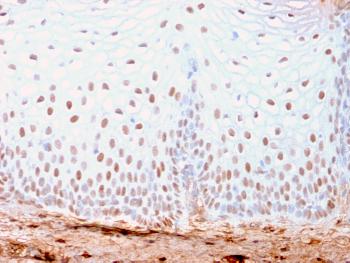
Recombinant human SPAM1 protein with C-terminal human Fc tag
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
| Amount : | 50 µg |
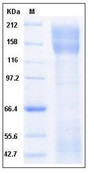
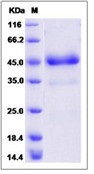
| Purification : | The purity of the protein is greater than 95% as determined by SDS-PAGE and Coomassie blue staining. |
| Content : | Lyophilized from sterile PBS, pH 7.4. Normally 5 % - 8 % trehalose is added as protectants before lyophilization. |
| Storage condition : | Store at -80°C for 12 months (Avoid repeated freezing and thawing) |
| Alternative Name : | HEL-S-96n; HYA1; HYAL1; HYAL3; HYAL5; PH-20; PH20; SPAG15 |
Expression Host : HEK293
The protein has a predicted molecular mass of 75.57 kDa after removal of the signal peptide.
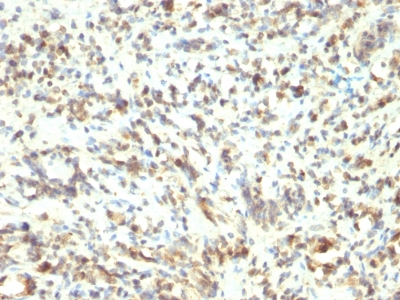
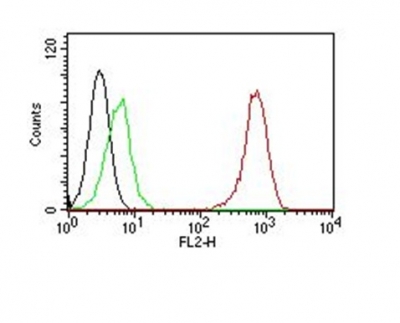
Hyaluronidase degrades hyaluronic acid, a major structural proteoglycan found in extracellular matrices and basement membranes. Six members of the hyaluronidase family are clustered into two tightly linked groups on chromosome 3p21.3 and 7q31.3. This gene was previously referred to as HYAL1 and HYA1 and has since been assigned the official symbol SPAM1; another family member on chromosome 3p21.3 has been assigned HYAL1. This gene encodes a GPI-anchored enzyme located on the human sperm surface and inner acrosomal membrane. This multifunctional protein is a hyaluronidase that enables sperm to penetrate through the hyaluronic acid-rich cumulus cell layer surrounding the oocyte, a receptor that plays a role in hyaluronic acid induced cell signaling, and a receptor that is involved in sperm-zona pellucida adhesion. Abnormal expression of this gene in tumors has implicated this protein in degradation of basement membranes leading to tumor invasion and metastasis. Multiple transcript variants encoding different isoforms have been found for this gene.
The protein has a predicted molecular mass of 75.57 kDa after removal of the signal peptide.
Hyaluronidase degrades hyaluronic acid, a major structural proteoglycan found in extracellular matrices and basement membranes. Six members of the hyaluronidase family are clustered into two tightly linked groups on chromosome 3p21.3 and 7q31.3. This gene was previously referred to as HYAL1 and HYA1 and has since been assigned the official symbol SPAM1; another family member on chromosome 3p21.3 has been assigned HYAL1. This gene encodes a GPI-anchored enzyme located on the human sperm surface and inner acrosomal membrane. This multifunctional protein is a hyaluronidase that enables sperm to penetrate through the hyaluronic acid-rich cumulus cell layer surrounding the oocyte, a receptor that plays a role in hyaluronic acid induced cell signaling, and a receptor that is involved in sperm-zona pellucida adhesion. Abnormal expression of this gene in tumors has implicated this protein in degradation of basement membranes leading to tumor invasion and metastasis. Multiple transcript variants encoding different isoforms have been found for this gene.
|
There are currently no product reviews
|














.png)