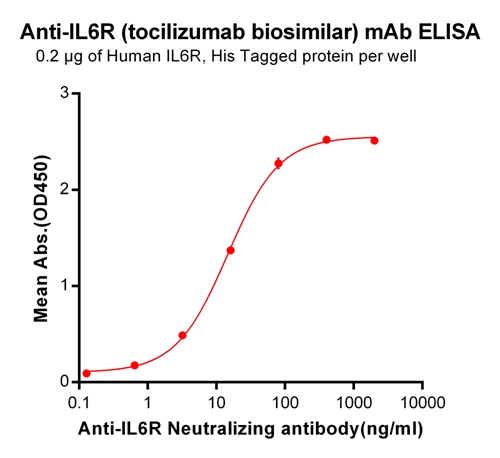
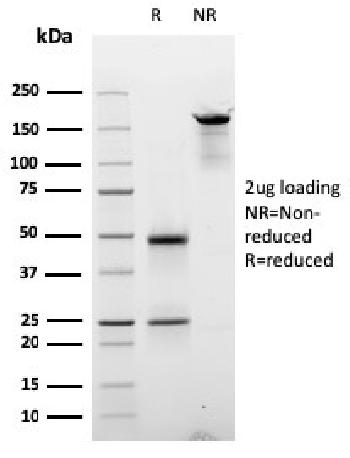
Anti-Human CD49D (Integrin alpha 4) (Natalizumab) – PE
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
| Amount : | 50 µg |
| Isotype : | Human IgG1k |
| Content : | Concentration : 0.2 mg/ml This R-phycoerythrin (R-PE) conjugate is formulated in 0.01 M phosphate buffered saline (150 mM NaCl) PBS pH 7.4, 1% BSA and 0.09% sodium azide as a preservative. |
| Storage condition : | This R-phycoerythrin (R-PE) conjugate is stable when stored at 2-8°C. Do not freeze. |
| Alternative Name : | CD49D; alpha 4 subunit of VLA-4 receptor; ITGA4; Integrin alpha-IV |
| Immunogen Information : | RAMOS cell line injected into mice. |
Expression Host : HEK-293
This non-therapeutic biosimilar antibody uses the same variable region sequence as the therapeutic antibody Natalizumab. Natalizumab binds to the alpha 4 subunit of alpha4beta1 and alpha4beta7 integrins. This product is for research use only.
Natalizumab is characterized as a disease-modifying therapy for multiple sclerosis (a disease of the central nervous system (CNS)), and inflammatory bowel disease. It works by inhibiting the migration of leukocytes to inflammation sites. The VCAM-1 and alpha4beta1-integrin interaction is necessary for leukocyte adhesion, firm attachment, and transmigration across the blood-brain barrier into the CNS. Natalizumab, a recombinant, humanized antibody, binds to alpha4beta1 -integrin and blocks its interaction with VCAM-1. Hence, leukocyte migration into brain tissue is inhibited, thereby reducing inflammation and preventing the formation of multiple sclerosis lesions.1 Inflammation in the gut pertaining to inflammatory bowel disease can be controlled in a similar fashion. Blocking alpha4beta7-integrin with a humanized, monoclonal antibody, specific to the alpha4beta7 heterodimer inhibits the migration of leukocytes into the inflamed intestinal tissue, thus, reducing inflammation in the gut.2 This cost-effective, research-grade Anti-Human CD49D (Natalizumab) utilizes the same variable regions from the therapeutic antibody Natalizumab making it ideal for research projects.
The suggested concentration for Natalizumab biosimilar antibody for staining cells in flow cytometry is <= 1.0 µg per 106 cells in a volume of 100 µl. Titration of the reagent is recommended for optimal performance for each application.
For Research Use Only. Not for use in diagnostic/therapeutics procedures.
|
There are currently no product reviews
|













.png)