Noggin Human, Sf9 Active
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
| Amount : | 10 µg |
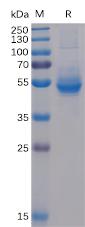
| Purification : | Greater than 95.0% as determined by:(a) Analysis by RP-HPLC (b) Analysis by SDS-PAGE. |
| Content : | Lyophilized from a 0.2µm filtered concentrated solution in PBS, pH7.4 and 0.02 % Tween-20 and 5% trehalose. It is recommended to reconstitute the lyophilized Noggin in sterile 18M Omega -cm H2O not less than 100µg/ml, which can then be further diluted to other aqueous solutions. |
| Storage condition : | Lyophilized Noggin although stable at room temperature for 3 weeks, should be stored desiccated below -18°C. Upon reconstitution Noggin should be stored at 4°C between 2-7 days and for future use below -18°C.Please prevent freeze-thaw cycles. |
| AA sequence : | QHYLHIRPAP SDNLPLVDLI EHPDPIFDPK EKDLNETLLR SLLGGHYDPG FMATSPPEDR PGGGGGAAGG AEDLAELDQL LRQRPSGAMP SEIKGLEFSE GLAQGKKQRL SKKLRRKLQM WLWSQTFCPV LYAWNDLGSR FWPRYVKVGS CFSKRSCSVP EGMVCKPSKS VHLTVLRWRC QRRGGQRCGW IPIQYPIISE CKCSC. |
| Alternative Name : | SYM1, SYNS1, NOG. |
Source: Sf9, Baculovirus cells.
Sterile Filtered White lyophilized (freeze-dried) powder.
Nogginwhich is encoded by the NOG gene, binds and inactivates members of the transforming growth factor-beta (TGF-beta) superfamily signaling proteins, such as bone morphogenetic protein-4 (BMP4). By diffusing through extracellular matrices more efficiently than members of the TGF-beta superfamily, noggin may play and important role in creating morphogenic gradients. Noggin appears to have pleiotropic effect, both early in development as well as in later stages. Noggin was originally isolated from Xenopus based on its ability to restore normal dorsal-ventral body axis in embryos that had been artificially ventralized by UV treatment. The results of the mouse knockout of noggin suggest that it is involved in numerous developmental processes, such as neural tube fusion and joint formation. There are several dominant human NOG mutations in unrelated families with proximal symphalangism (SYM1) and multiple synostoses syndrome (SYNS1). All NOG mutations altered evolutionarily conserved amino acid residues. The amino acid sequence of human noggin is highly homologous to that of Xenopus, rat and mouse.
Noggin produced in Sf9 Baculovirus cells is a glycosylated homodimer containing 205 amino acids and having a molecular mass of 47.9kDa under non-reducing conditions. (Molecular size on SDS-PAGE will appear at approximately 50-80kDa).Noggin is purified by proprietary chromatographic techniques.
Sterile Filtered White lyophilized (freeze-dried) powder.
Nogginwhich is encoded by the NOG gene, binds and inactivates members of the transforming growth factor-beta (TGF-beta) superfamily signaling proteins, such as bone morphogenetic protein-4 (BMP4). By diffusing through extracellular matrices more efficiently than members of the TGF-beta superfamily, noggin may play and important role in creating morphogenic gradients. Noggin appears to have pleiotropic effect, both early in development as well as in later stages. Noggin was originally isolated from Xenopus based on its ability to restore normal dorsal-ventral body axis in embryos that had been artificially ventralized by UV treatment. The results of the mouse knockout of noggin suggest that it is involved in numerous developmental processes, such as neural tube fusion and joint formation. There are several dominant human NOG mutations in unrelated families with proximal symphalangism (SYM1) and multiple synostoses syndrome (SYNS1). All NOG mutations altered evolutionarily conserved amino acid residues. The amino acid sequence of human noggin is highly homologous to that of Xenopus, rat and mouse.
Noggin produced in Sf9 Baculovirus cells is a glycosylated homodimer containing 205 amino acids and having a molecular mass of 47.9kDa under non-reducing conditions. (Molecular size on SDS-PAGE will appear at approximately 50-80kDa).Noggin is purified by proprietary chromatographic techniques.
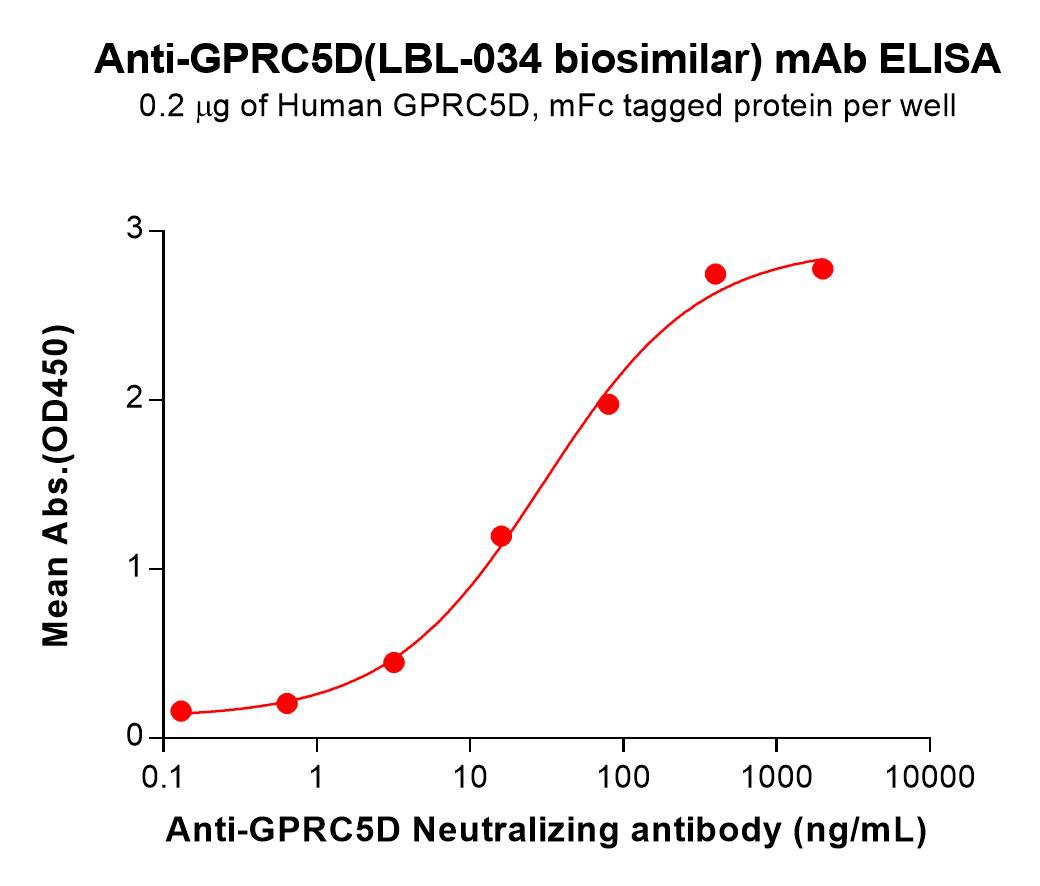
Measured by its ability to inhibit BMP-4-induced alkaline phosphatase production by ATDC5 mouse chondrogenic cellsans was fount to be 0.04-0.2 µg/mL in the presence of 50 ng/mL of Recombinant Human BMP-4.
For Research Use Only. Not for use in diagnostic/therapeutics procedures.
|
There are currently no product reviews
|














.png)








