Annexin V-FITC Apoptosis Detection Kit
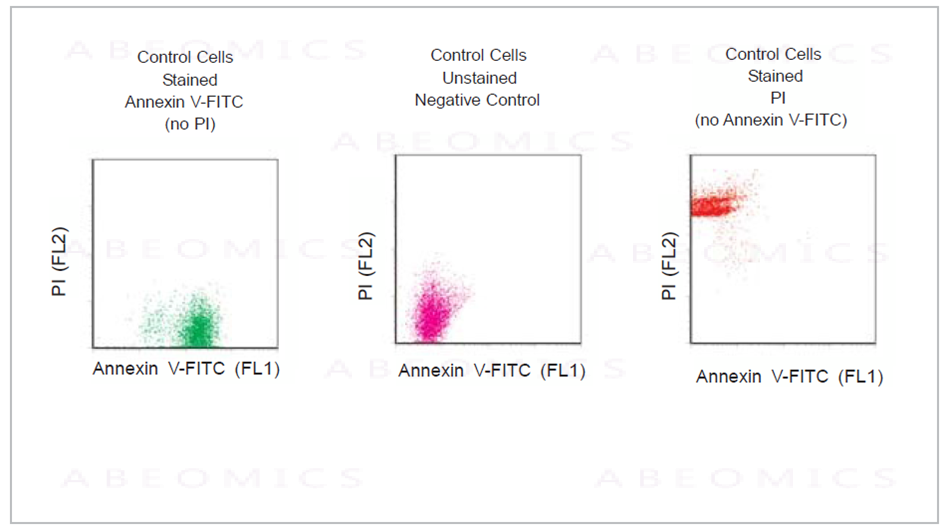
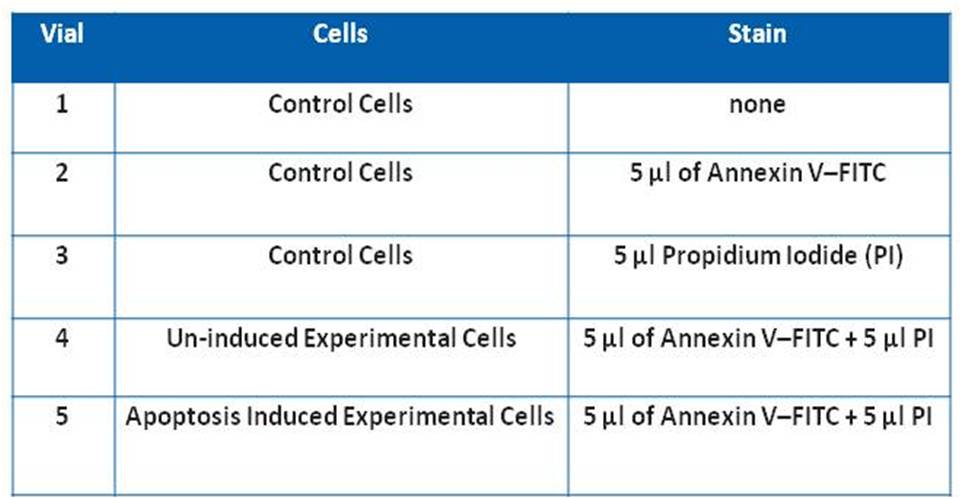
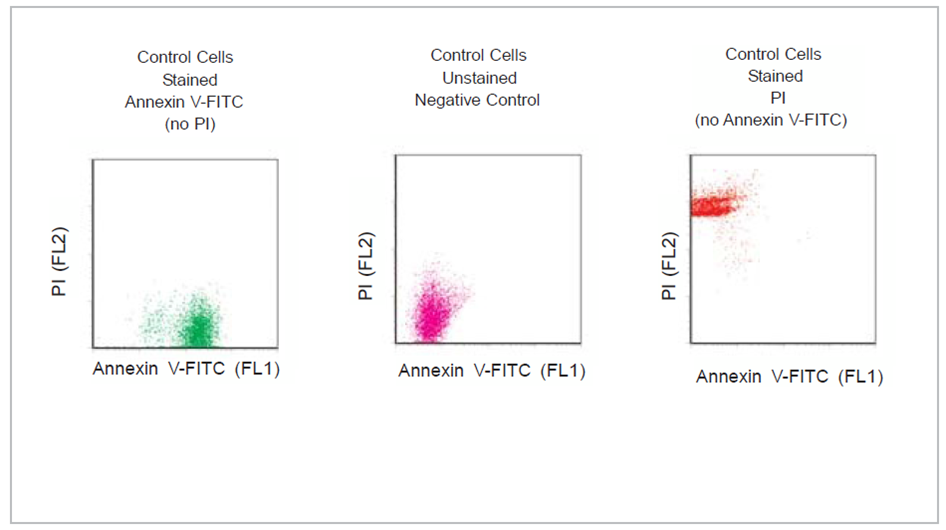
Fig.1: Control Cells were stained or unstained as indicated using the Annexin V-FITC Apoptosis Detection Kit.
Roll over image to zoom in
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
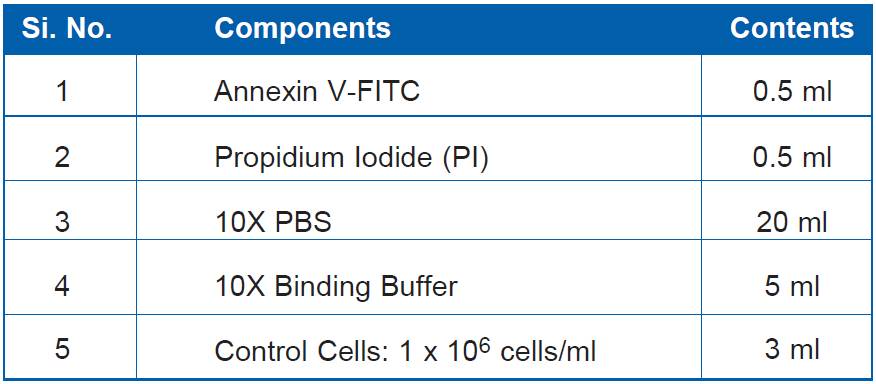
| Amount : | 100 Tests |
| Storage condition : | Store all kit components at 4°C. |
| Alternative Name : | , |
Apoptosis or programmed cell death is an evolutionary conserved, intrinsic program of cell death that occurs in various physiological and pathological situations. It is a key regulator of physiological growth control and regulation of tissue homeostasis. Apoptosis is considered a vital component of various processes including normal cell turnover, proper development and functioning of the immune system, hormone-dependent atrophy, embryonic development and chemical-induced cell death. The term apoptosis (a-po-toe-sis) was first used in a now-classic paper by Kerr, Wyllie, and Currie in 1972 to describe a morphologically distinct form of cell death, although certain components of the apoptosis concept had been explicitly described many years previously. Apoptosis is characterized by specific morphological changes of the dying cells; namely, loss of membrane asymmetry, cytoskeleton remodeling, plasma membrane blebbing, loss of the mitochondrial membrane potential, caspase activation, chromatin condensation, and DNA fragmentation.
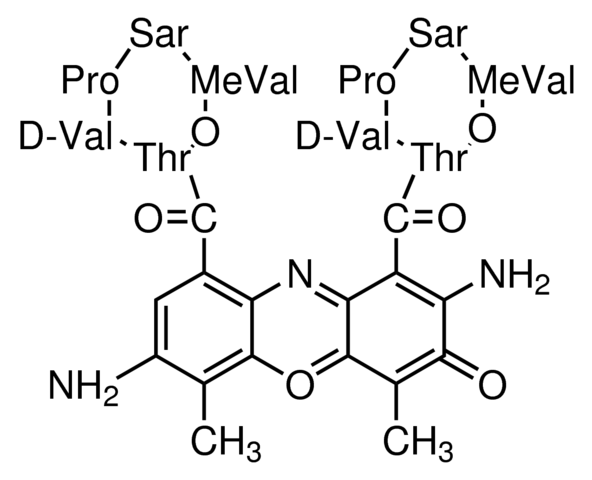
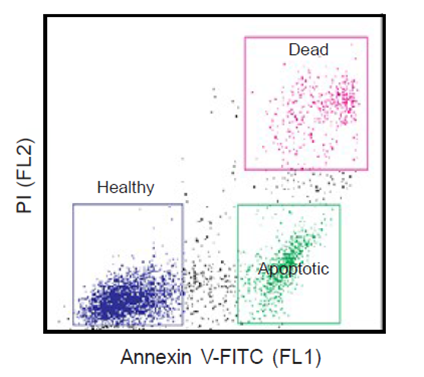
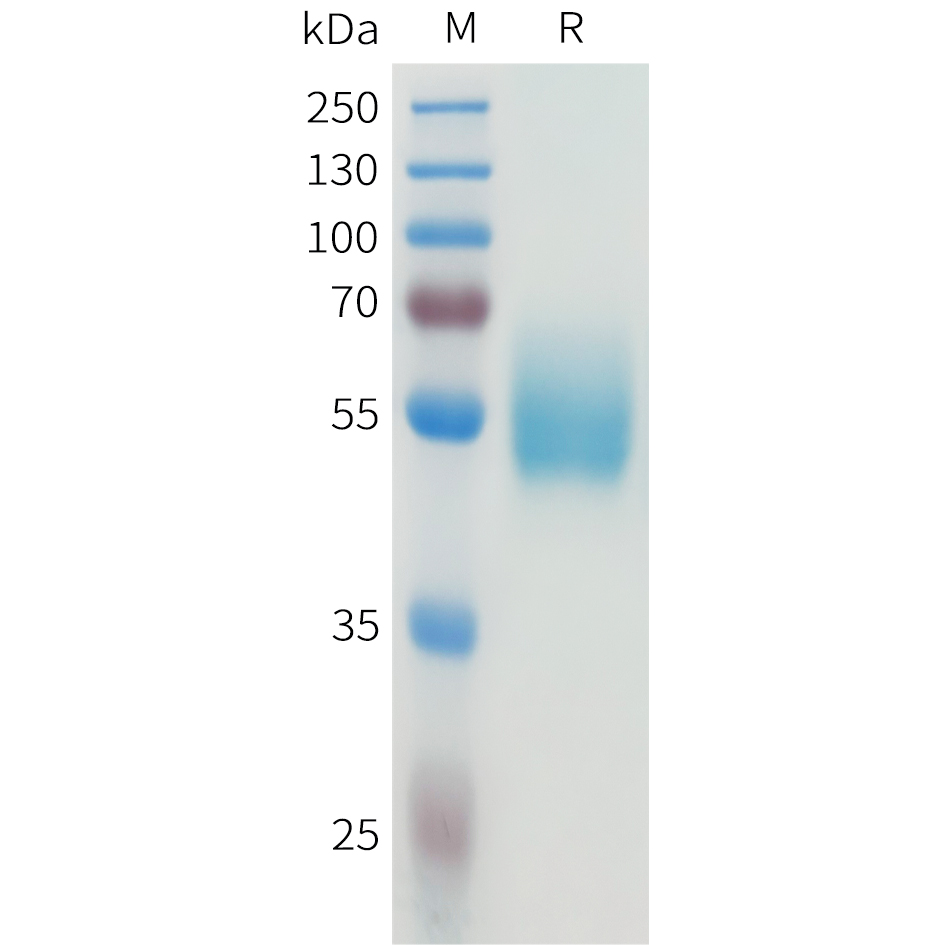
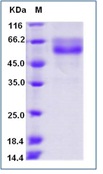
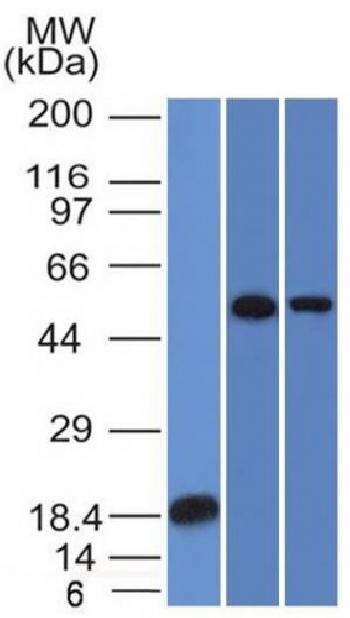
During apoptosis, the asymmetric distribution of phospholipids of the plasma membrane gets lost and phosphatidylserine (PS) is translocated to the outer leaflet of the plasma membrane. There, PS acts as one major “eat me” signal that ensures efficient recognition and uptake of apoptotic cells by phagocytes. Once on the cell surface, PS can be easily detected by staining with a fluorescent conjugate of Annexin V, a 35-36 kDa Ca2+ dependent phospholipid-binding protein that has a high affinity for PS, and binds to cells with exposed PS. The one-step staining procedure takes only 10 minutes. Detection can be analyzed by flow cytometry or by fluorescence microscopy. Annexin V may be conjugated to fluorochromes including FITC. This format retains its high affinity for PS and thus serves as a sensitive probe for flow cytometric analysis of cells that are undergoing apoptosis.
For Research Use Only. Not for use in diagnostic/therapeutics procedures.
1. Paul P, Verma S, Kumar Panda P, Jaiswal S, Sahu BR, Suar M. Molecular insight to influential role of Hha-TomB toxin-antitoxin system for antibacterial activity of biogenic silver nanoparticles. Artif Cells Nanomed Biotechnol. 2018;46(sup3):S572-S584. doi: 10.1080/21691401.2018.1503598. Epub 2018 Nov 16. PubMed PMID: 30444141.
2. Anuja K, Chowdhury AR, Saha A, Roy S, Rath AK, Kar M, Banerjee B. Radiation-induced DNA damage response and resistance in colorectal cancer stem-like cells. Int J Radiat Biol. 2019 Jun;95(6):667-679. doi: 10.1080/09553002.2019.1580401. Epub 2019 Mar 20. PubMed PMID: 30753097.
3. Maity P, Bepari M, Pradhan A, Baral R, Roy S, Maiti Choudhury S. Synthesis and characterization of biogenic metal nanoparticles and its cytotoxicity and anti-neoplasticity through the induction of oxidative stress, mitochondrial dysfunction and apoptosis. Colloids Surf B Biointerfaces. 2018 Jan 1;161:111-120. doi: 10.1016/j.colsurfb.2017.10.040. Epub 2017 Oct 13. PubMed PMID: 29055863.
4. Banerjee A, Patra SA, Sahu G, Sciortino G, Pisanu F, Garribba E, Carvalho MFNN, Correia I, Pessoa JC, Reuter H, Dinda R.
A Series of Non-Oxido VIV Complexes of Dibasic ONS Donor Ligands: Solution Stability, Chemical Transformations, Protein Interactions, and Antiproliferative Activity.
Inorg Chem. 2023 May 22;62(20):7932-7953. doi: 10.1021/acs.inorgchem.3c00753. Epub 2023 May 8.
PMID: 37154533; PMCID: PMC10367067.
Amol S. Shinde & Rita R. Lala
Mannose-anchored solid lipid nanoparticles loaded with atorvastatin calcium and vinpocetine as targeted therapy for breast cancer
5. Future Journal of Pharmaceutical Sciences volume 9, Article number: 81 (2023)
https://fjps.springeropen.com/articles/10.1186/s43094-023-00531-y
|
There are currently no product reviews
|













.png)