Anti-Human CD279 (PD-1) (Nivolumab) – APC
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
| Amount : | 50 µg |
| Content : | Concentration : 0.2 mg/ml This Allophycocyanin (APC) conjugate is formulated in 0.01 M phosphate buffered saline (150 mM NaCl) PBS pH 7.4, 1% BSA and 0.09% sodium azide as a preservative. |
| Storage condition : | This Allophycocyanin (APC) conjugate is stable when stored at 2-8°C. Do not freeze. |
| Alternative Name : | PD1; PD-1; CD279; SLEB2; hPD-1; hPD-l; hSLE1 |
| Immunogen Information : | Human PD-1 |
Expression Host : HEK-293
This non-therapeutic biosimilar antibody uses the same variable region sequence as the therapeutic antibody Nivolumab. Clone 5C4.B8 binds to the extracellular portion of Human/Cynomolgus PD-1 and does not bind to other IgG superfamily proteins. This product is for research use only.
Programmed cell death protein 1 (PD-1) is a protein on the surface of cells that plays a role in the maintenance of self-tolerance. PD-1 promotes self-tolerance via the down-regulation of the immune system which results in the suppression of T cell inflammatory activity. PD-L1 and PD-L2 are the two ligands known to bind PD-1. PD-L1 has increased expression in several cancers. PD-L2 has a more limited expression and is primarily expressed by dendritic cells and only some tumor lines. Inhibition of the interaction of PD-1 with its ligands can function as an immune checkpoint blockade through the improvement of In vitro T-cell responses and via the mediation of anti-tumor activity. Nivolumab disrupts the negative signal that is responsible for T-cell activation and proliferation by binding to PD-1 on activated immune cells to selectively block the interaction of the PD-1 receptor with its ligands. Emerging research suggests that combined blockade of PD-1 and CTLA-4, with nivolumab and ipilimumab respectively, could produce greater antitumor activity than blockade of either pathway alone. This cost-effective, research-grade Anti-Human CD279 (PD-1) (Nivolumab) utilizes the same variable regions from the therapeutic antibody Nivolumab making it ideal for research projects.
Endotoxin : <= 1.0 EU/mg as determined by the LAL method
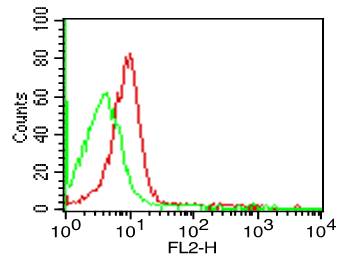
The suggested concentration for Nivolumab biosimilar antibody for staining cells in flow cytometry is <= 1.0 µg per 106 cells in a volume of 100 µl. Titration of the reagent is recommended for optimal performance for each application.
For Research Use Only. Not for use in diagnostic/therapeutics procedures.
|
There are currently no product reviews
|













.png)








