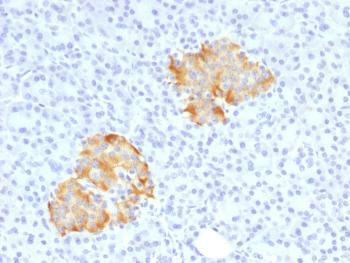
Recombinant human HLA-A protein with C-terminal human Fc tag
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
| Amount : | 50 µg |
| Purification : | The purity of the protein is greater than 95% as determined by SDS-PAGE and Coomassie blue staining. |
| Content : | Lyophilized from sterile PBS, pH 7.4. Normally 5 % - 8 % trehalose is added as protectants before lyophilization. |
| Storage condition : | Store at -80°C for 12 months (Avoid repeated freezing and thawing) |
| Alternative Name : | Aw-33; Aw-74; HLAA |
Expression Host : HEK293
The protein has a predicted molecular mass of 56.76 kDa after removal of the signal peptide.
HLA-A belongs to the HLA class I heavy chain paralogues. This class I molecule is a heterodimer consisting of a heavy chain and a light chain (beta-2 microglobulin). The heavy chain is anchored in the membrane. Class I molecules play a central role in the immune system by presenting peptides derived from the endoplasmic reticulum lumen. They are expressed in nearly all cells. The heavy chain is approximately 45 kDa and its gene contains 8 exons. Exon 1 encodes the leader peptide, exons 2 and 3 encode the alpha1 and alpha2 domains, which both bind the peptide, exon 4 encodes the alpha3 domain, exon 5 encodes the transmembrane region, and exons 6 and 7 encode the cytoplasmic tail. Polymorphisms within exon 2 and exon 3 are responsible for the peptide binding specificity of each class one molecule. Typing for these polymorphisms is routinely done for bone marrow and kidney transplantation. Hundreds of HLA-A alleles have been described.
The protein has a predicted molecular mass of 56.76 kDa after removal of the signal peptide.
HLA-A belongs to the HLA class I heavy chain paralogues. This class I molecule is a heterodimer consisting of a heavy chain and a light chain (beta-2 microglobulin). The heavy chain is anchored in the membrane. Class I molecules play a central role in the immune system by presenting peptides derived from the endoplasmic reticulum lumen. They are expressed in nearly all cells. The heavy chain is approximately 45 kDa and its gene contains 8 exons. Exon 1 encodes the leader peptide, exons 2 and 3 encode the alpha1 and alpha2 domains, which both bind the peptide, exon 4 encodes the alpha3 domain, exon 5 encodes the transmembrane region, and exons 6 and 7 encode the cytoplasmic tail. Polymorphisms within exon 2 and exon 3 are responsible for the peptide binding specificity of each class one molecule. Typing for these polymorphisms is routinely done for bone marrow and kidney transplantation. Hundreds of HLA-A alleles have been described.
|
There are currently no product reviews
|













.png)