Human IL-35 (Interleukin 35) Pre-Coated ELISA Kit
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
| Amount : | 1 × 96 Tests |
| Content : | 1 × 96 well Format (96 tests) |
| Storage condition : | Please refer to the Manual |
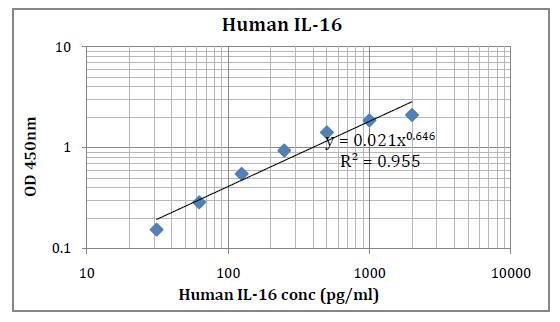
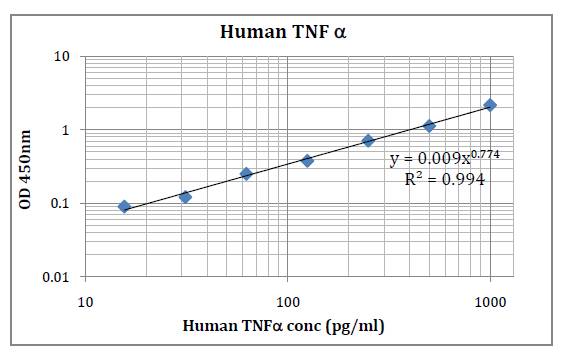
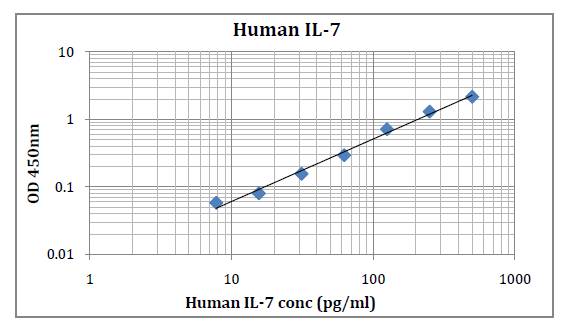
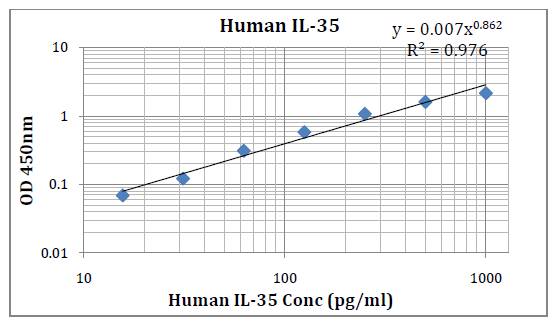
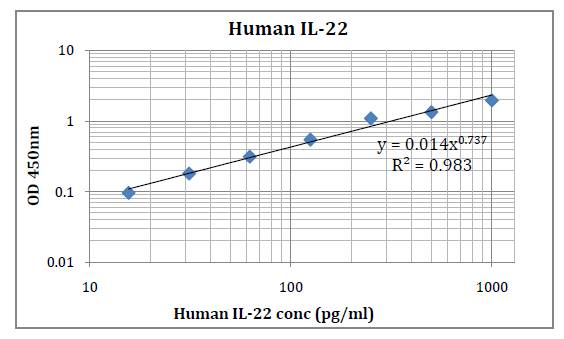
This kit was based on sandwich enzyme-linked immune-sorbent assay technology. Anti-Human IL-35 antibody was pre-coated into 96-well plates. Biotin conjugated anti-human IL-35 detection antibody was used. Standards, test samples and biotin conjugated detection antibody were added to the wells subsequently. Wash buffer was used to wash any non-specific binding. HRP conjugated Streptavidin was used as secondary antibody. TMB substrates were used to visualize HRP enzymatic reaction. TMB was catalyzed by HRP to produce a blue color product that changed into yellow after adding acidic stop solution. The density of yellow is proportional to the Human IL-35 amount of samples captured in the plate. Optical Density (O.D) can be read at absorbance 450 nm in a microplate reader. Concentration of Human IL-35 can be calculated using the standard curve.
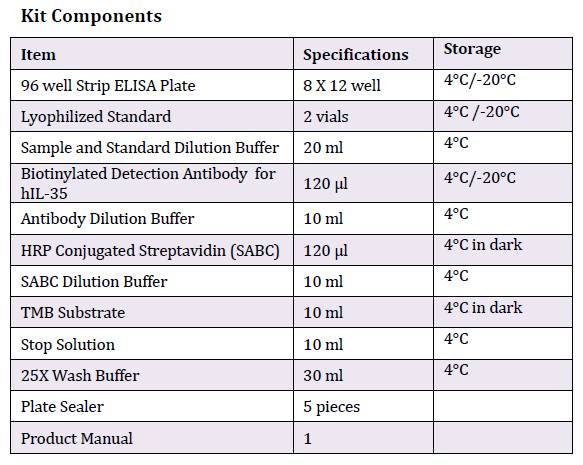
This immunoassay kit allows for the in vitro quantitative determination of Human IL-35 concentrations in serum, plasma and other biological fluids.
Detection Range: 15.6 – 1000 pg/ml
Sensitivity: < 9.4 pg/ml
For Research Use Only. Not for use in diagnostic/therapeutics procedures.
Su Y, Feng S, Luo L, Liu R, Yi Q. Correction to: Association between IL-35 and coronary arterial lesions in children with Kawasaki disease. Clin Exp Med. 2018 Aug 31. doi: 10.1007/s10238-018-0525-2. [Epub ahead of print] PubMed PMID: 30171456.
|
There are currently no product reviews
|













.png)