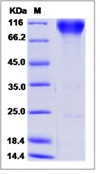
Recombinant Human Endothelial Cell Adhesion Molecule/PECAM-1/CD31 (C-Fc)
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
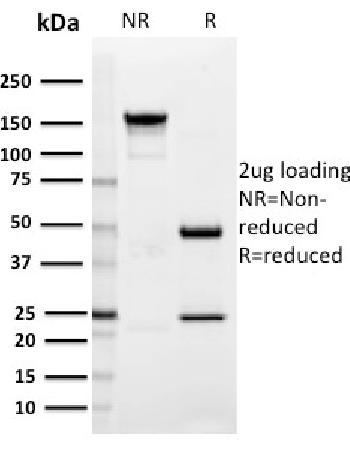
| Amount : | 50 µg |
| Content : | Lyophilized from a 0.2 µm filtered solution of PBS, pH7.4. |
| AA sequence : | Recombinant Human Platelet Endothelial Cell Adhesion Molecule is produced by our Mammalian expression system and the target gene encoding Gln28-Lys601 is expressed with a Fc tag at the C-terminus. |
| Alternative Name : | Platelet endothelial cell adhesion molecule; PECAM-1; EndoCAM; GPIIA; PECA1; CD31; PECAM1 |
Source : Human Cells;
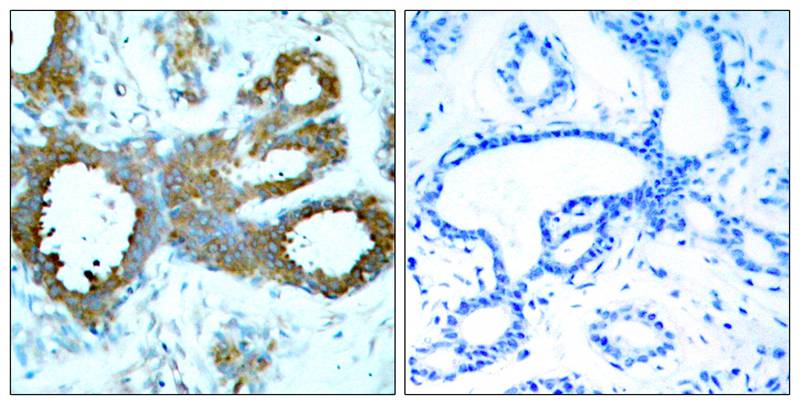
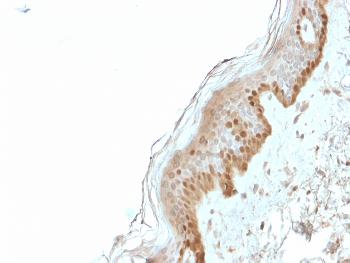
Platelet Endothelial Cell Adhesion Molecule (PECAM1, CD31), is a heavily glycosylated transmembrane protein belonging to the immunoglobulin (Ig) superfamily of cell adhesion molecules. CD31 is composed of an extracellular domain (ECD) of 574 amino acids (aa) containing six Ig-like domains, a transmembrane domain, and a 118 aa cytoplasmic domain. CD31 is highly expressed on endothelial cells and at a lower level on platelets, granulocytes, macrophages, dendritic cells, T and B cells, and natural killer (NK) cells. It is involved in cell adhesion and is required for transepithelial migration of leukocytes (TEM). CD31 acts as a homophilic receptor through its extracellular domain and is involved in downstream signaling via its cytoplasmic domain. This domain contains highly conserved ITIM motifs which, once tyrosine phosphorylated, recruit and activate the signaling molecules Src and SHP2. The resulting inhibition of TCR signaling increases the activation threshold of T cells, thus reinforcing peripheral tolerance and preventing development of autoimmunity. CD31 additionally regulates immune responses by acting as a key inhibitory receptor in dendritic cell development. CD31 is required for the transendothelial migration of leukocytes through intercellular junctions of vascular endothelial cells.
Platelet Endothelial Cell Adhesion Molecule (PECAM1, CD31), is a heavily glycosylated transmembrane protein belonging to the immunoglobulin (Ig) superfamily of cell adhesion molecules. CD31 is composed of an extracellular domain (ECD) of 574 amino acids (aa) containing six Ig-like domains, a transmembrane domain, and a 118 aa cytoplasmic domain. CD31 is highly expressed on endothelial cells and at a lower level on platelets, granulocytes, macrophages, dendritic cells, T and B cells, and natural killer (NK) cells. It is involved in cell adhesion and is required for transepithelial migration of leukocytes (TEM). CD31 acts as a homophilic receptor through its extracellular domain and is involved in downstream signaling via its cytoplasmic domain. This domain contains highly conserved ITIM motifs which, once tyrosine phosphorylated, recruit and activate the signaling molecules Src and SHP2. The resulting inhibition of TCR signaling increases the activation threshold of T cells, thus reinforcing peripheral tolerance and preventing development of autoimmunity. CD31 additionally regulates immune responses by acting as a key inhibitory receptor in dendritic cell development. CD31 is required for the transendothelial migration of leukocytes through intercellular junctions of vascular endothelial cells.
|
There are currently no product reviews
|

















.png)







